Will Cu ions show ESR spectrum?
Will Cu ions show ESR spectrum?
Cu2+, Ag2+, and Au2+ in their 2Σg+ ground states have been observed in solid neon matrices at 4 K via X-band electron-spin-resonance (ESR) spectroscopy.
How do you calculate EPR spectrum?
Table 2.
- The number of lines from the hyperfine interaction can be determined by the formula: 2NI + 1.
- For example, for •CH3, the radical’s signal is split to 2NI+1= 2*3*1/2+1=4 lines, the ratio of each line’s intensity is 1:3:3:1.
What is the geometry of copper 2 complex?
Cu(I) is a d10, closed shell ion, and since the most common coordination geometry is four, the preferred complex geometry will be tetrahedral. On the other hand Cu(II), with a d9 configuration, has square planar or octahedral geometry.
Is EPR copper active?
The d9 configuration of Cu2+ means that its compounds are paramagnetic making EPR of Cu(II) containing species a useful tool for both structural and mechanistic studies.
Are Cu2+ complexes paramagnetic?
Under this condition, Cu2+ion have one unpaired 4Pz electron. Hence Cu2+ion show paramagnetic properties.
Which reference standard is used in ESR spectroscopy?
The most widely used reference is 1,1-diphenyl-2-picrylhydrazyl free radical (DPPH) which is completely in free radical state and its g value is 2.0036. The reference substance is placed along with the unknown in the same dual resonant cavity.
What is the G value in EPR?
The g-value of a free electron is 2.0023, and the g-values of most free radicals are very close to this value, since the unpaired electron has very little orbital contribution to the magnetic moment.
What is the g-factor in EPR?
The g-factor of an EPR sample determines the position in the magnetic field (at a given microwave frequency) where an EPR transition will occur. g-factor indicates field position for resonance. The g-factor is a constant but the field for resonance changes with microwave frequency.
What are the geometry of complexes with coordination number 2 4 and 6?
The Structures of Complexes
| Coordination Number | Molecular Geometry | Example |
|---|---|---|
| 4 | square planar (d8) | [NiCl4]2− |
| 5 | trigonal bipyramidal | [CoCl5]2− |
| 5 | square pyramidal | [VO(CN)4]2− |
| 6 | octahedral | [CoCl6]3− |
Why EPR is more sensitive than NMR?
The primary advantage of EPR over nuclear magnetic resonance (NMR) in the study of defects is that of sensitivity. This higher sensitivity is mainly a result of the larger quantum of energy absorbed when an electron rather than a nucleus is flipped in a given magnetic field.
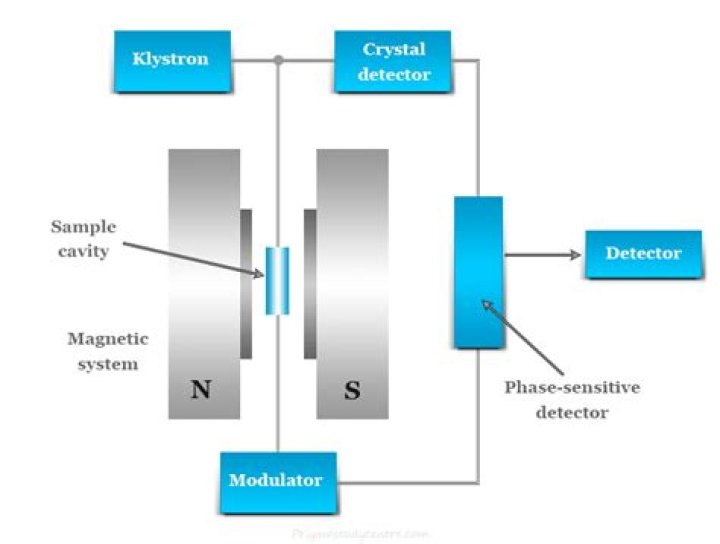
How does EPR spectroscopy work?
In EPR spectrometers a phase-sensitive detector is used. As mentioned earlier, an EPR spectrum is obtained by holding the frequency of radiation constant and varying the magnetic field. Absorption occurs when the magnetic field “tunes” the two spin states so that their energy difference is equal to the radiation.



