What type of bond is po4 3?
What type of bond is po4 3?
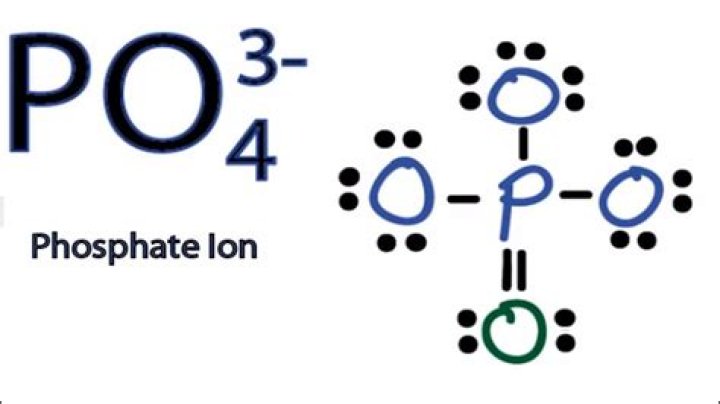
Lewis structure of PO43- ion In the lewis structure of PO43-, three is a double bond between phosphorous atom and one oxygen atom. Between other oxygen atoms, there are only single bonds with phosphorous atom. Also, each oxygen atom has a -1 charge.
How is po4 3 formed?
The overall charge on the phosphate ion is -3 and the phosphorus atom is in an oxidation state of +5. In the Lewis structure of PO43-, P forms single bonds with 3 oxygen atoms and forms a double bond with one oxygen atom. These oxygen atoms carry a charge of -1.
How many bonding electrons are present in PO43?
In the Lewis structure of PO43- there are a total of 32 valence electrons.
What is the the shape molecular geometry of PO43 -?
3−
| Central atom: | P |
|---|---|
| Total VSEP: | 8 |
| 4 x double bonds | − 4 pairs |
| Revised Total: | 4 |
| Geometry: | Tetrahedral |
What bond is PO4?
a phosphate group is a polyatomic ion which contains both ionic and covalent bonds. By definition, a phosphate group is PO4 with a charge of -3.
Is PO4 3 covalent or ionic?
The bonds in PO4^3- have high covalent character… 32% ionic character and 68% covalent character. “PO4” isn’t a compound, it is the phosphate ion, and has an oxidation state of -3.
What is a PO4 3?
PO43- is a chemical derivative of phosphoric acid with a chemical name Phosphate. Phosphate is also called Phosphate ion or Orthophosphate. It is a trivalent inorganic anion and a conjugate base of hydrogen phosphate. and positively charged ions such as calcium or sodium; another group composed of esters.
What type of ion is PO4 3?
phosphate ion
Phosphate
| PubChem CID | 1061 |
|---|---|
| Description | Phosphate(3-) is a phosphate ion that is the conjugate base of hydrogenphosphate. It is a phosphate ion and a trivalent inorganic anion. It is a conjugate base of a hydrogenphosphate. ChEBI Inorganic salts of phosphoric acid. Medical Subject Headings (MeSH) |
How many bonding electrons does PO4 3 have?
32 valence
Video: Drawing the Lewis Structure for PO43- In the Lewis structure of PO43- there are a total of 32 valence electrons. For the Lewis structure for PO4 3- you should take formal charges into account to find the best Lewis structure for the molecule. Remember, PO4 3- has a negative three charge on the molecule.
What PO4 3?
PO43- is a chemical derivative of phosphoric acid with a chemical name Phosphate. Phosphate is also called Phosphate ion or Orthophosphate. It is a trivalent inorganic anion and a conjugate base of hydrogen phosphate. Phosphate is found in many phosphate minerals.
Is PO4 3 polar or nonpolar?
This means there is no net dipole in the molecule, making it non-polar. Hence, PO43- is a non-polar molecule. Polar molecules must contain polar bonds due to a difference in electronegativity between the bonded atoms.



