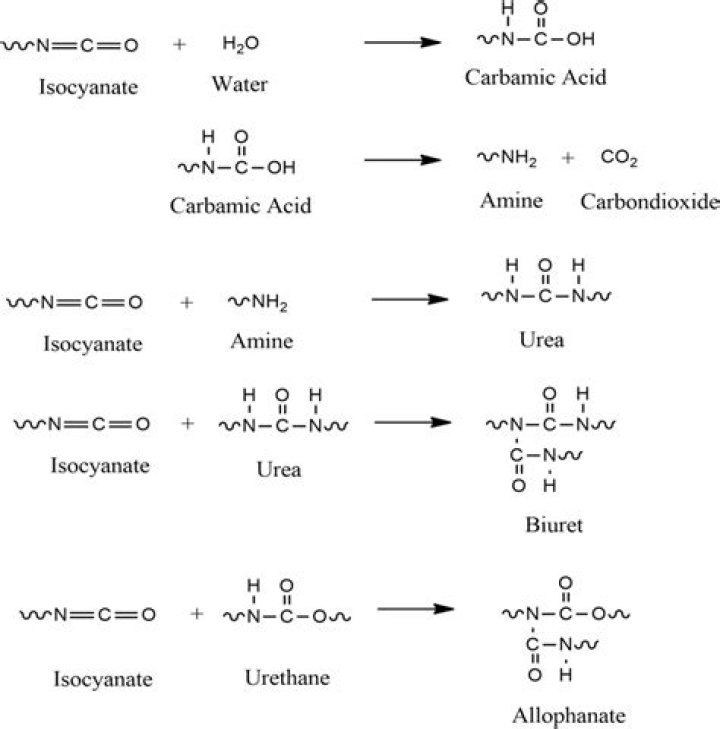
What is the product of isocyanate reacts with amine?
Isocyanates also react with amines to give ureas: R2NH + R’NCO → R2NC(O)N(H)R’
Why are isocyanates so reactive?
An active hydrogen atom-containing nucleophilic center attacks the electrophilic carbon atom and active hydrogen is added to the nitrogen atom. NCO-bonded electron acceptor groups increase the reactivity and donor groups reduce it. For that reason, aromatic isocyanates are more reactive than aliphatic ones.
Is isocyanate electron withdrawing?
It consists of two electron-withdrawing components, the chlorosulfonyl group (SO2Cl) and the isocyanate group (-N=C=O).
How do you quench an isocyanate?
isopropyl alcohol to neutralize the isocyanate, then wash thoroughly with soap and water.
What is isocyanate used for?
Isocyanates are widely used in manufacturing materials like polyurethane foams, rubbers, plastics, varnishes, adhesives and paints. A list of the most widely used industrial organic isocyanates and their main uses is in Table 1.
What is the difference between cyanate and isocyanate?
In context|chemistry|lang=en terms the difference between isocyanate and cyanate. is that isocyanate is (chemistry) the univalent radical -n=c=o. while cyanate is (chemistry) to treat or react with cyanide.
What are the effects of isocyanate?
Health effects of isocyanate exposure include irritation of skin and mucous membranes, chest tightness, and difficult breathing. Isocyanates include compounds classified as potential human carcinogens and known to cause cancer in animals.
How do you test for isocyanate poisoning?
There are no routine clinical blood or urine tests for isocyanates. Test inhalation challenge to isocyanate is not advised except in experienced laboratories owing to the danger of severe asthma attack.
What is polymeric isocyanate?
Isocyanates are compounds containing the isocyanate group (-NCO). They react with compounds containing alcohol (hydroxyl) groups to produce polyurethane polymers, which are components of polyurethane foams, thermoplastic elastomers, spandex fibers, and polyurethane paints.
What is thionyl chloride used for?
Thionyl chloride is mainly used in the industrial production of organochlorine compounds, which are often intermediates in pharmaceuticals and agrichemicals.
Is MDI an isocyanate?
Methylene diphenyl diisocyanate (MDI) is an aromatic diisocyanate. The 4,4′ isomer is most widely used, and is also known as 4,4′-diphenylmethane diisocyanate. This isomer is also known as Pure MDI. MDI reacts with polyols in the manufacture of polyurethane.
How do you identify an isocyanate?
Isocyanates are in the hardener of many autobody paints Autobody paints usually contain hexamethylene diisocyanate (HDI) and/or isophorone diisocyanate (IPDI). Look for the word “isocyanate” or these chemical names on a product label or MSDS to determine if a product you are using contains isocyanates.