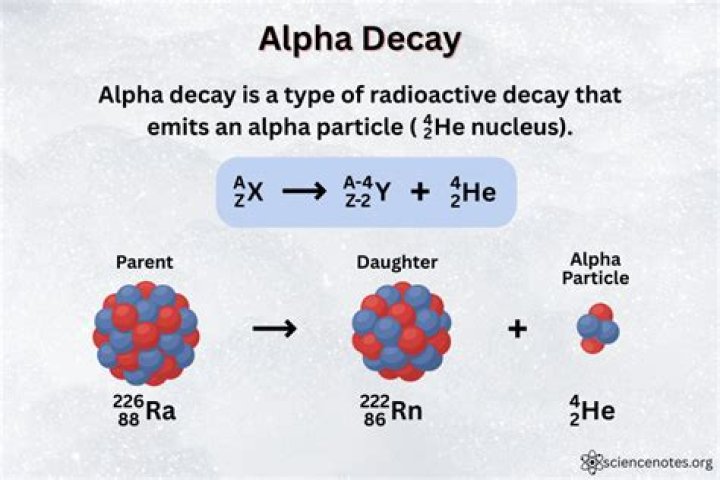
What is the product of alpha decay of curium 243?
Curium-243 undergoes α decay to plutonium- 239 : 243Cm⟶239Pu+α
How long is a half-life for curium 246?
Isotopes of curium
| Isotope | ||
|---|---|---|
| abundance | half-life (t1/2) | |
| 245Cm | syn | 8500 y |
| 246Cm | syn | 4730 y |
| 247Cm | syn | 1.56×107 y |
How many neutrons does curium 242 have?
Curium 242 is an isotope of the chemical element curium. It has atomic number 96 and mass number 242. That means, there are 96 protons in the atomic nucleus of the curium atom along with 146 neutrons.
How does plutonium-239 decay?
Plutonium-239 decays through spontaneous fission while emitting alpha particles.
What happens when plutonium-239 decays?
For example, the decay products of plutonium-238 and plutonium-239 are uranium- 234 and uranium-235. The decay process continues until a stable, non-radioactive decay product is formed. Radiation is released during the decay process in the form of alpha and beta particles and gamma radiation.
What is curium used for?
Curium has two main uses: as a fuel for Radioisotope Thermal Generators (RTGs) on board satellites, deep space probes, planetary surface rovers and in heart pacemakers, and as a alpha emitter for alpha particle X-Ray spectrometry, again particularly in space applications.
What compound is curium in?
Scientists have produced several curium compounds. They include: curium dioxide (CmO2), curium trioxide (Cm2O3), curium bromide (CmBr3), curium chloride (CmCl3), curium chloride (CmCl3), curium tetrafluoride (CmF4) and curium iodide (CmI3).
When was curium 242 synthesized?
1944
It was first synthesized in 1944 by Glenn T. Seaborg and his team at the University of California in Berkeley using the reaction 239Pu (4He, n) 242Cm. The element was named after Pierre and Marie Curie, who discovered radium and polonium.
Why was curium named?
Curium is named in honour of Pierre and Marie Curie, who pioneered the study of radioactivity in the final days of the 19th century. Nineteen radioisotopes of curium are known to exist, the first of which, 242Cu was isolated in the hydroxide form in 1947 and in its elemental form in 1951.
What is curium half life?
about 15,600,000 years
Isotopes of curium Though none are stable, 247Cm has a half-life of about 15,600,000 years. It decays into 243Pu through alpha decay. Cm has a half-life of 163 days and decays into 238Pu through alpha decay or decays through spontaneous fission.
How is curium synthesized?
Curium can be made in very small amounts by the neutron bombardment of plutonium in a nuclear reactor. Minute amounts may exist in natural deposits of uranium. Only a few grams are produced each year.