What are the units for the molar mass of a gas?
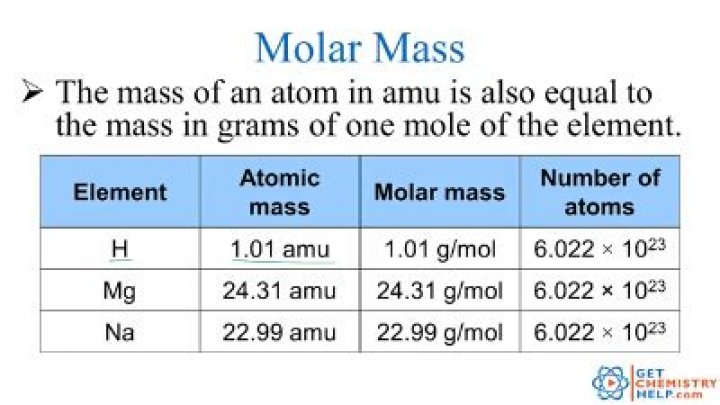
Molar mass (M) is equal to the mass of one mole of a particular element or compound; as such, molar masses are expressed in units of grams per mole (g mol–1) and are often referred to as molecular weights.
How do you find the molar mass of a gas using the ideal gas law?
First the ideal gas law will be used to solve for the moles of unknown gas (n). Then the mass of the gas divided by the moles will give the molar mass. Step 2: Solve. Now divide g by mol to get the molar mass.
What are the units for the ideal gas law?
In SI units, p is measured in pascals, V is measured in cubic metres, n is measured in moles, and T in kelvins (the Kelvin scale is a shifted Celsius scale, where 0.00 K = −273.15 °C, the lowest possible temperature). R has the value 8.314 J/(K⋅mol) ≈ 2 cal/(K⋅mol), or 0.0821 L⋅atm/(mol⋅K).
What is the formula unit of molar mass?
grams per mole
The molar mass of a substance is defined as the mass of 1 mol of that substance, expressed in grams per mole, and is equal to the mass of 6.022 × 10 23 atoms, molecules, or formula units of that substance.
What is the value of R in PV mRT?
units as: R = 8.314 J/mol·K. This means that for air, you can use the value R = 287 J/kg·K. If you use this value of R, then technically the formula should be written as pV = mRT, where m represents the mass of air in kg (and we avoid having to do any calculations with moles.)
How do you calculate ideal gas law?
The ideal gas law formula states that pressure multiplied by volume is equal to moles times the universal gas constant times temperature….Ideal Gas Law Formula
- P = pressure.
- V = volume.
- n = number of moles.
- T = temperature.
- R = gas constant.
What is ideal gas equation class 11?
(b) It is the combination of Boyle’s law, Charles’s law and Avogadro’s law PV/T = constant the value of constant depends on for amount of gas and the units in which pressure and volume are measured. (c) PV = nRT. PV =m/M × RT. The equation is called as an ideal gas equation.
How is formula unit mass calculated?
To calculate the formula unit mass, find out the number of atoms of every element present in the chemical formula. Add together the relative atomic mass values for all the atoms of every element present.
How do you calculate formula units?
There are 6.022×1023 in 1 mole of anything, including formula units. You need to determine the number of moles in 0.335 g CaO . Once you know the number of moles of CaO , you can determine the number of formula units by multiplying the number of moles by 6.022×1023 .
What is the value of R in SI unit?
8.314JK
Where P is pressure of the gas, V is Volume, n- no. Of moles, R is a universal gas constant which has constant value and T represents temperature of the given conditions. The value of R in SI units is 8.314JK−1mol−1.