What amino acids have primary amines?
What amino acids have primary amines?
Primary amines exist at the N-terminus of each polypeptide chain and in the side-chain of lysine (Lys, K) amino acid residues.
What are the primary amine?
Primary amine (1o amine): An amine in which the amino group is directly bonded to one carbon of any hybridization which cannot be a carbonyl group carbon. General primary amine molecular structure. X = any atom but carbon; usually hydrogen.
Is amine reactive?
The reactivity of amines is similar to ammonia: amines are basic, nucleophilic, and react with alkyl halides, acid chlorides, and carbonyl compounds. Additionally, aromatic amines are highly reactive in electrophilic aromatic substitution.
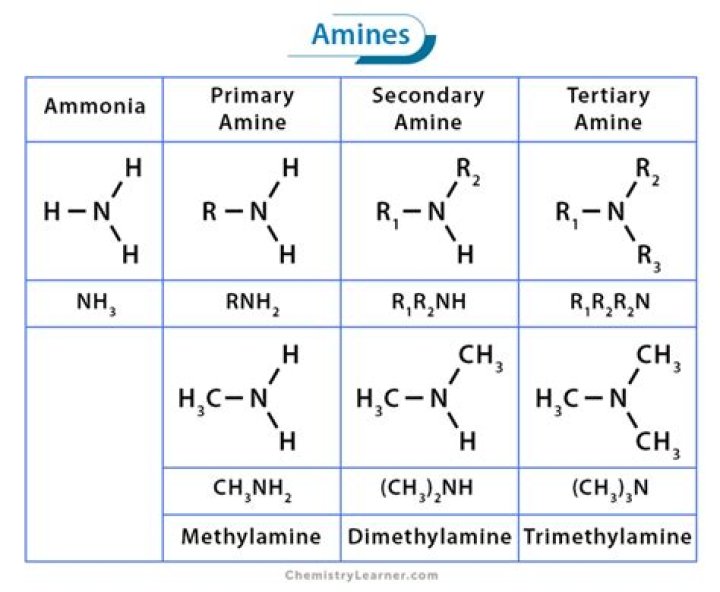
Is Dimethylamine a primary secondary or tertiary amine?
Primary (1°) amines—Primary amines arise when one of three hydrogen atoms in ammonia is replaced by an alkyl or aromatic group. Important representatives include dimethylamine, while an example of an aromatic amine would be diphenylamine. Tertiary (3°) amines—In tertiary amines, nitrogen has three organic substituents.
Is aniline primary amine?
Abstract. Aniline (benzenamine) is the simplest of the primary aromatic amines. Aromatic amines can be produced by reduction of the corresponding nitro compound, the ammonolysis of an aromatic halide or phenol, and by direct amination of the aromatic ring.
What are primary secondary and tertiary amines give one example of each?
Amines are classified as primary, secondary, or tertiary according to the number of carbons bonded directly to the nitrogen atom. Primary amines have one carbon bonded to the nitrogen. Secondary amines have two carbons bonded to the nitrogen, and tertiary amines have three carbons bonded to the nitrogen.
Which amine is more basic primary secondary or tertiary?
The small amount of extra negative charge built up on the nitrogen atom makes the lone pair even more attractive towards hydrogen ions. Correspondingly, primary, secondary, and tertiary alkyl amines are more basic than ammonia.