Why is asymmetric stretching higher than symmetric stretching?
Symmetric stretching involves the movement of atoms vibration or stretching of two bonds in and out together simultaneously and thus, there is no change in dipole moment in symmetric stretching. So, asymmetric stretching appears at a higher wavenumber and absorbs or needs more energy than symmetric stretching.
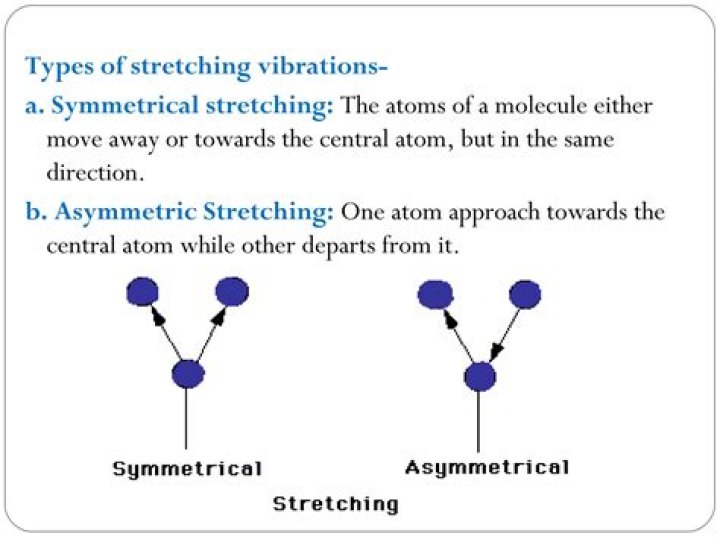
What is symmetrical stretching?
Symmetric stretching: Simultaneous vibration of two bonds, in which the bonds elongate together and contract together.
What is asymmetric stretching?
In an asymmetric stretch, they are going in opposite directions. One bond gets longer as the other gets shorter, so the change in dipole moment is much less. It takes energy to separate positive and negative charges from each other, so the symmetric vibration has a higher frequency (energy).
What is the difference between symmetrical and asymmetrical molecules?
A symmetrical molecule is one whose appearance does not change if you turn it about an axis of symmetry; original and rotated states are indistinguishable from one another. By contrast, an asymmetrical molecule has no axis of symmetry; you can tell if it has been rotated.
Is asymmetric stretching IR active?
The asymmetric stretch of carbon dioxide is IR active because there is a change in the net molecular dipole (Figure 5. 2). In the asymmetric stretch, one bond is stretched and is now more polarizable while the other bond is compressed and is less polarizable.
Is symmetric stretch IR active?
The symmetric stretch does not change the dipole moment so it is not IR active.
Is symmetric stretching IR active?
Why is symmetric stretch IR inactive?
Some kinds of vibrations are infrared inactive. The stretching vibrations of completely symmetrical double and triple bonds, for example, do not result in a change in dipole moment, and therefore do not result in any absorption of light (but other bonds and vibrational modes in these molecules do absorb IR light).
What is difference between symmetric and asymmetric carbon?
If carbon is bonded with the same atoms, then it is said to be a symmetric carbon atom. Thus, this carbon atom is called a symmetric carbon atom. Now, if a carbon atom is bonded with all different atoms or groups of atoms. Then such a carbon atom is called an asymmetric carbon atom.
How does symmetry affect IR spectrum?
Symmetry is one of the factors that can significantly reduce the number of bands observed in the infrared. If stretching a bond does not cause a change in the dipole moment, the vibration will not be able to interact with the infrared radiation and the vibration will be infrared inactive.
What’s the difference between asymmetric and chiral?
An asymmetric carbon atom (chiral carbon) is a carbon atom that is attached to four different types of atoms or groups of atoms. Molecules that cannot be superimposed on their own mirror image are said to be chiral like mirror image.
What are asymmetric Centres?
Filters. (chemistry) An atom having a spatial arrangement of ligands which is not superposable on its mirror image. noun.