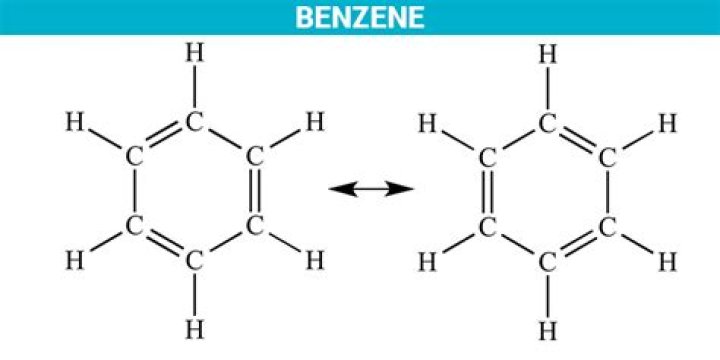
Why benzene is called aromatic?
Originally, benzene was considered aromatic because of its smell: it has an “aromatic” odor. It is now considered aromatic because it obeys Hückel’s rule: 4n+2 = number of π electrons in the hydrocarbon, where n must be an integer. In the case of benzene, we have 3 π bonds (6 electrons), so 4n+2=6 .
Is benzene aliphatic or aromatic?
Complete answer:
| Aliphatic functional groups | Aromatic functional groups |
|---|---|
| Examples for aliphatic compounds are methane, propane, butane etc. | Examples for aromatic compounds are benzene, toluene etc. |
What is difference between aromatic and benzene?
As nouns the difference between aromatic and benzene is that aromatic is a fragrant plant or spice added to a dish to flavour it while benzene is (organic compound) an aromatic hydrocarbon of formula c6h6 whose structure consists of a ring of alternate single and double bonds.
Is benzene aromatic and unsaturated?
Aromatic hydrocarbons are a special class of unsaturated hydrocarbon based on a six carbon ring moiety called benzene.
What do you mean by aromatic compound?
Aromatic compounds are chemical compounds that consist of conjugated planar ring systems accompanied by delocalized pi-electron clouds in place of individual alternating double and single bonds. They are also called aromatics or arenes. The best examples are toluene and benzene.
Is benzene an aromatic compound?
Benzene (C6H6) is the best-known aromatic compound and the parent to which numerous other aromatic compounds are related.
Which is composed of aromatic hydrocarbons?
Monocyclic aromatic hydrocarbons (MAHs) are composed of, a single aromatic ring. Benzene, toluene, ethylbenzene and xylenes (BTEX) are well-known environmental pollutants which are the most volatile and water-soluble aromatic hydrocarbons.
What are some examples of aromatic compounds?
Typical examples of aromatic compounds are benzene, naphthalene, and anthracene.
What are examples of benzene?
Examples of simple benzene derivatives are phenol, toluene, and aniline, abbreviated PhOH, PhMe, and PhNH2, respectively. Linking benzene rings gives biphenyl, C6H5–C6H5. Further loss of hydrogen gives “fused” aromatic hydrocarbons, such as naphthalene, anthracene, phenanthrene, and pyrene.
Why are they called aromatic hydrocarbons?
Aromatic compounds, originally named because of their fragrant properties, are unsaturated hydrocarbon ring structures that exhibit special properties, including unusual stability, due to their aromaticity. They are often represented as resonance structures containing single and double bonds.
What is the purpose of benzene?
Benzene is a widely used industrial chemical. Benzene is found in crude oil and is a major part of gasoline. It’s used to make plastics, resins, synthetic fibers, rubber lubricants, dyes, detergents, drugs and pesticides. Benzene is produced naturally by volcanoes and forest fires.