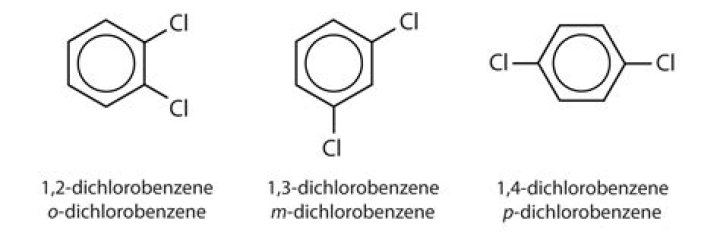
Which of the following is the alternative name for 1/3-dichlorobenzene?
1,3-Dichlorobenzene
| PubChem CID | 10943 |
|---|---|
| Structure | Find Similar Structures |
| Chemical Safety | Laboratory Chemical Safety Summary (LCSS) Datasheet |
| Molecular Formula | C6H4Cl2 |
| Synonyms | 1,3-DICHLOROBENZENE m-Dichlorobenzene 541-73-1 Benzene, 1,3-dichloro- m-Phenylene dichloride More… |
What is another name for 1/2 dichlorobenzene?
ortho-dichlorobenzene
1,2-Dichlorobenzene, also named ortho-dichlorobenzene, is an organic compound. It is a non-polar colorless liquid that is miscible in most organic solvents. This derivative of benzene differs from the parent compound by the presence of two adjacent chlorine atoms.
What is P-dichlorobenzene used for?
Paradichlorobenzene is used as a fumigant insecticide to control clothes moths. It is also found in deodorant blocks made for trash cans and toilets. Paradichlorobenzene was first registered for use in the United States in 1942, and it is sometimes called 1,4-dichlorobenzene.
What does P-dichlorobenzene look like?
P-dichlorobenzene appears as a white colored liquid with the odor of moth balls. Denser than water and insoluble in water.
Which axis is are present in p-dichlorobenzene?
1,4-Dichlorobenzene D. 2h D2h point groups contain two C2 axes perpendicular to the main rotation axis with one σh plane.
How many isomers are there for dichlorobenzene?
three isomeric
dichlorobenzene, any of three isomeric organohalogen compounds known as 1,2-, 1,3-, or 1,4-dichlorobenzene (also called ortho-, meta-, and para-dichlorobenzene, respectively).
What is dichlorobenzene d4?
1,4-Dichlorobenzene (1,4-DCB, p-DCB, or para-dichlorobenzene, sometimes abbreviated as PDCB or para) is an organic compound with the formula C6H4Cl2. The molecule consists of a benzene ring with two chlorine atoms (replacing hydrogen atoms) on opposing sites of the ring.
How do I get rid of dichlorobenzene?
You can directly remove it by column chromatrography using hexane as eluent and then adding a more polar solvent to get your ketonic product.
Is dichlorobenzene safe for humans?
* Breathing 1,4-Dichlorobenzene can irritate the nose and throat causing coughing and wheezing. * Exposure can cause headache, dizziness, swelling around the eyes, nausea and vomiting. * 1,4-Dichlorobenzene may be a CARCINOGEN in humans since it has been shown to cause kidney and liver cancer in animals.
What is the dipole moment of p-dichlorobenzene?
zero
Answer: Dipole moment of p – dichlorobenzene is zero due to symmetrical structure.
What is the molar mass of P-dichlorobenzene?
147.01
Other names – 1,4-Dichlorobenzene
| C6H4Cl2 | Para dichlorobenzene |
|---|---|
| Density | 1.25 g/cm3 |
| Molecular Weight/ Molar Mass | 147.01 |
| Boiling Point | 174 °C |
| Melting Point | 53.5 °C |