Which is the example of aziridine class agent?
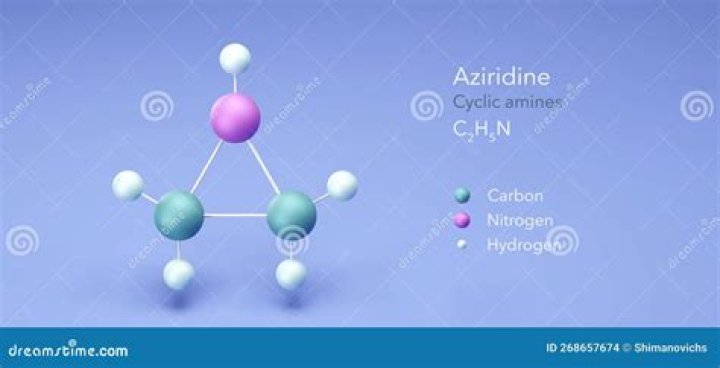
Which is the example of aziridine class agent?
The biological properties of aziridine-containing compounds such as azinomycins, mitomycins, FR-900482, ficellomycin, miraziridine, maduropeptin, and azicemicins are of significant interest. The antibiotic and antitumor properties of several of these compounds are well known.
Which drugs belong to aziridine ring?
Several drugs feature aziridine rings, including mitomycin C, porfiromycin, and azinomycin B (carzinophilin).
What is the structure of aziridine?
Aziridine is an organic compound consisting of the three-membered heterocycle (CH2)2NH. It is a colorless, toxic, volatile liquid that is of significant practical interest. Aziridine was discovered in 1888 by the chemist Siegmund Gabriel.
Why is aziridine a weak base?
Aziridine is less basic than acyclic aliphatic amines, with a pKa of 7.9 for the conjugate acid, due to the increased s character of the nitrogen electron pair. The weak basicity of aziridine and its derivatives is ascribed to the strain in the 3-membered ring compound.
Why is aziridine highly reactive?
Aziridines are three-membered cyclic amines (azacyclopropanes). The ring-strain associated with these molecules makes them extremely reactive compounds. The presence of a carboxylate group or a vinylic group on the ring-carbon atom(s), and an arylsulfonyl group on the ring-nitrogen atom further activates the ring.
Is aziridine a weak base?
Aziridine (C2H5N), a weak base, reacts with a strong acid such as HNO3 to form its conjugate acid aziridinium (C2H5NH ). A strong acid reacts completely with a stoichiometric amount of weak base after each addition of the strong acid.
Is aziridine an amine?
Aziridines are three-membered cyclic amines (azacyclopropanes). The ring-strain associated with these molecules makes them extremely reactive compounds.
What is the structure of oxirane?
C2H4O
Ethylene oxide/Formula