Which is Hofmann degradation reaction?
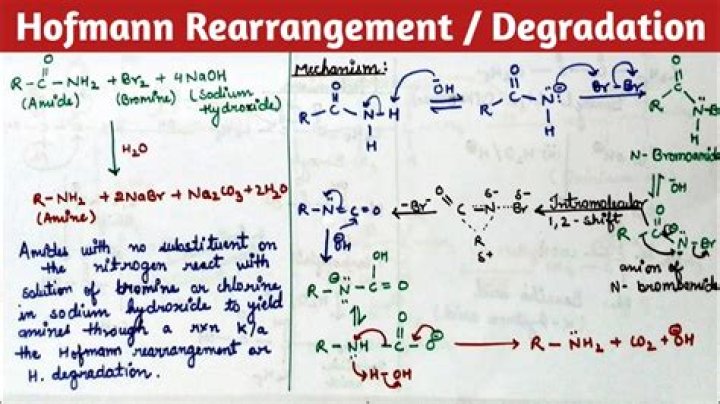
The Hofmann rearrangement (Hofmann degradation) is the organic reaction of a primary amide to a primary amine with one fewer carbon atom. The reaction involves oxidation of the nitrogen followed by rearrangement of the carbonyl and nitrogen to give an isocyanate intermediate.
What is Hoffmans Bromamide degradation give example?
What Is Hoffmann bromamide Reaction? When an amide is treated with bromine in an aqueous or ethanolic solution of sodium hydroxide, degradation of amide takes place leading to the formation of primary amine. This reaction involving degradation of amide and is popularly known as Hoffmann bromamide degradation reaction.
What is Hofmann degradation amides?
Hofmann rearrangement, also known as Hofmann degradation and not to be confused with Hofmann elimination, is the reaction of a primary amide with a halogen (chlorine or bromine) in strongly basic (sodium or potassium hydroxide) aqueous medium, which converts the amide to a primary amine.
Why Gabriel phthalimide synthesis is not used for the preparation of aniline?
Can aniline be prepared by Gabriel phthalimide synthesis? Gabriel’s synthesis can not prepare aniline. Using this process, aniline can not be prepared, because aryl halides do not undergo nucleophilic substitution with the phthalimide-formed anion.
What is Hofmann alkaloid degradation?
amines. quaternary ammonium compounds is the Hofmann degradation, which takes place when the hydroxides are strongly heated, generating a tertiary amine; the least-substituted alkyl group is lost as an alkene.
What is Hofmann Bromamide reaction?
Hoffmann’s bromamide reaction involves the conversion of a carboxylic acid amide into an amine with a loss of carbon atom. On treatment with aqueous sodium hypobromite, this Hoffmann’s reaction results in the shortening of a carbon chain.
Why is Hoffmann Bromamide a degradation reaction?
In Hoffmann bromamide degradation reaction, an amide reacts with bromine and aqueous solution of sodium hydroxide which produces primary amine. This a degradation reaction as primary amine in the product has one carbon lesser than primary amide (in the reactant).
Why is it called Hofmann degradation?
It is named after its discoverer, August Wilhelm von Hofmann. The reaction starts with the formation of a quaternary ammonium iodide salt by treatment of the amine with excess methyl iodide (exhaustive methylation), followed by treatment with silver oxide and water to form a quaternary ammonium hydroxide.
What Cannot be prepared by Gabriel phthalimide synthesis?
Hence, the correct answer is (d) iso-butyl amine. NOTE – The process of Gabriel phthalimide synthesis only works with primary alkyl halides/haloalkanes. Aromatic amines cannot also be prepared by this method.
Which of the following Cannot be prepared by Gabriel’s method?
Butylamine, isobutylamine and 2-phenyl ethylamine are primary amine hence these can be prepared by Gabriel’s synthesis but N-methyl benzylamine is a secondary amine and therefore, it cannot be prepared by Gabriel’s synthesis.
Where does Hofmann elimination occur?
The authors developed a two-compartment pharmacokinetic model in which hepatic and/or renal elimination occurs from the central compartment (Cl organ), and Hofmann elimination and ester hydrolysis occur from both central and peripheral compartments (Cl nonorgan).
Who discovered Hoffmann Bromamide?
August Wilhelm von Hofmann
Hoffmann Bromamide reaction is an organic reaction in which a primary amide is converted to a primary amine. After conversion, the primary amine has one less carbon atom. The reaction was discovered by August Wilhelm von Hofmann, and hence is named after him.