Which hybridization is present in ammonia?
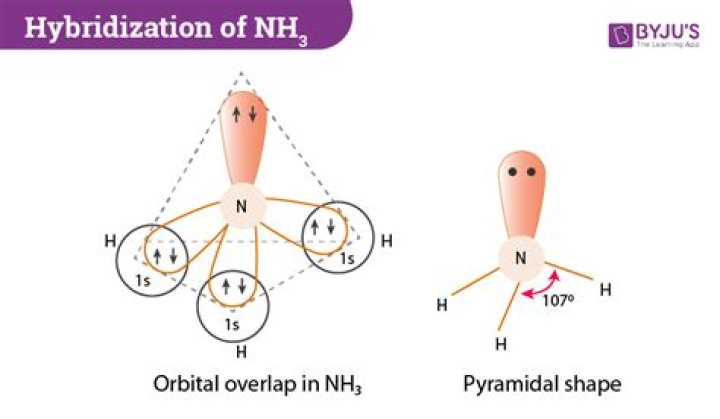
The type of hybridization present in ammonia (NH3) molecule is sp3.
What is the hybridization of sp3?
The term “sp3 hybridization” refers to the mixing character of one 2s-orbital and three 2p-orbitals to create four hybrid orbitals with similar characteristics. In order for an atom to be sp3 hybridized, it must have an s orbital and three p orbitals.
Does NH4+ have sp3 hybridization?
The NH4+ ion has no pi bonds. As a result, all four electrons contained in the atomic orbitals in the outermost shell of the nitrogen atom can participate in hybridization, making it SP3.
Is ammonia a sp2 or sp3?
Ammonia ( NH3) , or, more accurately, the central atom in ammonia, is sp3 hybridized.
What is the shape of ammonia?
trigonal pyramidal shape
The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom.
Is I3+ sp3 hybridized?
Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape. Since I3+ has sp3 hybridisation and two lone pair on central I atom therefore its shape would be Bent or V shape.
Is so3 sp3 hybridized?
SO3 has a sp2 type of hybridization. We will understand how the molecule obtains such hybridized state below….Hybridization of SO3 (Sulphur Trioxide)
| Name of the Molecule | Sulphur Trioxide |
|---|---|
| Molecular Formula | SO3 |
| Hybridization Type | sp2 |
| Bond Angle | 120o |
| Geometry | Trigonal Planar |
Is ammonia a sp3?
In the ammonia molecule (NH3), 2s and 2p orbitals create four sp3hybrid orbitals, one of which is occupied by a lone pair of electrons.
Why is NH3 sp3 not sp2?
Nitrogen has its 2s , 2px , 2py , and 2pz orbitals to use. Its 2pz orbital is aligned precisely along the z axis. Since ammonia is three-dimensional, it requires sp3 hybridization to make its bonds.
What is the shape and hybridization in NH3?
Hybridization of NH3 (Ammonia)
| Name of the Molecule | Ammonia |
|---|---|
| Molecular Formula | NH3 |
| Hybridization Type | sp3 |
| Bond Angle | 107o |
| Geometry | Pyramidal or Distorted Tetrahedral |