Which has the largest covalent radius?
Covalent Radius of the elements
| Helium | 28 pm | Iridium |
|---|---|---|
| Nitrogen | 71 pm | Silver |
| Carbon | 76 pm | Thallium |
| Boron | 85 pm | Ruthenium |
| Beryllium | 96 pm | Lead |
What is the covalent radius of SI?
116
Atomic radius
| atomic number | symbol | Covalent (single bond) |
|---|---|---|
| 12 | Mg | 139 |
| 13 | Al | 126 |
| 14 | Si | 116 |
| 15 | P | 111 |
What is covalent radius 11th?
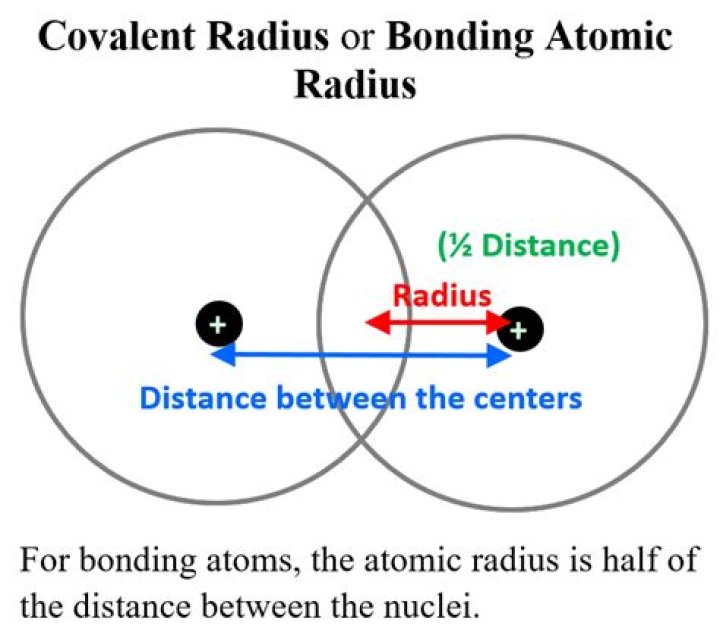
– Covalent radius: it is one half of the distance between the nuclei of two covalently bonded atoms in a homo-diatomic molecule which is called covalent radius atom. – Metallic radius: It is taken as half of the internuclear distance separating the metal cores in the metallic crystal.
How do you find the covalent radius of carbon?
Thus the covalent radius of carbon atom can be calculated by subtracting the covalent radius of Cl atom from d(C-Cl) bond length. The covalent radius of Cl atom can also be obtained, provided that covalent radius of C atom is known.
Which has minimum covalent radius?
So, $Ni$ has the least covalent radius among the following given elements and the correct option is, (D).
What is covalent and ionic radii?
Measurement of Radius Nonpolar atomic radii: The radius of an atom is derived from the bond lengths within nonpolar molecules; one-half the distance between the nuclei of two atoms within a covalent bond. Crystal Radii: The atomic or ionic radius is determined using electron density maps fro X-ray data.
How do you find covalent radius?
The covalent radius, rcov, is a measure of the size of an atom that forms part of one covalent bond. It is usually measured either in picometres (pm) or angstroms (Å), with 1 Å = 100 pm. In principle, the sum of the two covalent radii should equal the covalent bond length between two atoms, R(AB) = r(A) + r(B).
What is covalent radius Class 10?
The covalent radius of an element is considered to be one half of the covalent bond distance of a molecule where the atoms concerned are participating in single bonding. In other words, It may be defined as one-half of the distance between the centres of the nuclei of two similar atoms bonded by a single covalent bond.
How do you find the covalent radius?
What do you mean by covalent radius?
Why does boron form covalent compounds?
Hint: Boron is a chemical element with atomic number 5 which means it is small in size. Also the first three ionization enthalpies of boron are very high hence it cannot lose electrons to form a cation and therefore it shares electrons to form a covalent compound.
Is covalent radii greater than atomic radii?
Covalent Radius Covalent radii will increase in the same pattern as atomic radii. The reason for this trend is that the bigger the radii, the further the distance between the two nuclei.