Which amino acids are zwitterions?
Which amino acids are zwitterions?
At physiological pH, monoaminomonocarboxylic amino acids, e.g., glycine and alanine, exist as zwitterions.
Which amino acids are zwitterions at pH 7?
Glutamine is the only amino acid that is zwitterionic at pH 7. The structure of an amino acid is such that it allows it to behave as both, an acid and a base at a certain pH value.
What type of amino acid is lysine?
essential amino acid
an essential amino acid, has a positively charged ε-amino group (a primary amine). Lysine is basically alanine with a propylamine substituent on theβcarbon.
How do amino acids form Zwitterions?
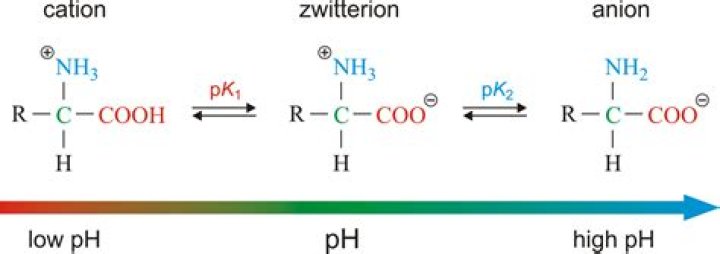
Zwitterions in simple amino acid solutions An amino acid has both a basic amine group and an acidic carboxylic acid group. There is an internal transfer of a hydrogen ion from the -COOH group to the -NH2 group to leave an ion with both a negative charge and a positive charge. This is called a zwitterion.
At what pH are amino acids zwitterions?
= 5.02
At pH = 5.02, the pH = pI so the amino acid will exist as the zwitterion with both the positive and negative charges as shown above.
At what pH is cysteine a zwitterion?
The structure of cysteine at pH = 7 shows that the side group is protonated. So we must conclude that even though the pKa is 8.33, the sulfhydryl (−SH) is acting as an acid. The isoelectric point, pI, is the pH at which the zwitterion is the dominant species.
What are called Zwitter ions Class 12?
Hint: Zwitterion , also known as the dipolar ion is formed in amino acids due to the transfer of the proton from the carboxylic acid group to the amino group and in this, the acidic nature is due to ammonium ion and basic nature is due to carboxylate ion.
What is lysine known for?
Lysine is an essential amino acid with many benefits that range from preventing cold sores to reducing anxiety and promoting wound healing. As a building block of proteins, it may also have numerous other benefits.
Is lysine a neutral amino acid?
There are three amino acids that have basic side chains at neutral pH. These are arginine (Arg), lysine (Lys), and histidine (His). Their side chains have carboxylic acid groups whose pKa’s are low enough to lose protons, becoming negatively charged in the process.