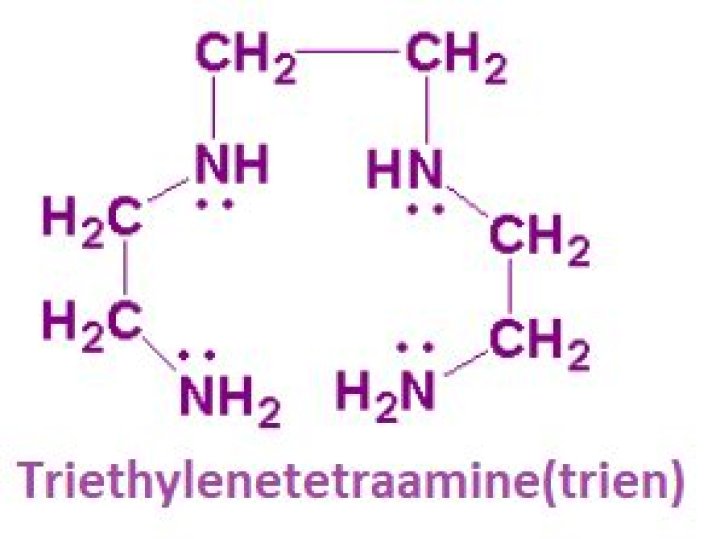
What is trien ligand?
TETA is a tetradentate ligand in coordination chemistry, where it is referred to as trien. Octahedral complexes of the type M(trien)L2 can adopt several diastereomeric structures.
What is triethylenetetramine used for?
Triethylenetetramine is a copper chelating agent used for the management of Wilson’s disease in cases where penicillamine therapy is clinically inappropriate. Triethylenetatramine (TETA) is a highly selective divalent Cu(II) chelator and orphan drug that revereses copper overload in tissues.
What is the molecular formula of triethylenetetramine?
C6H18N4
Triethylenetetramine/Formula
What is Trien Denticity?
Tetradentate ligands bind with four donor atoms, an example being triethylenetetramine (abbreviated trien). Hexadentate ligands bind with six atoms, an example being EDTA (although it can bind in a tetradentate manner). Ligands of denticity greater than 6 are well known.
Is triethylenetetramine a Tridentate?
The ligand triethylenetetramine is tetradentate.
What type of ligand is ethylenediamine?
bidentate ligand
– Ethylenediamine is an example of bidentate ligand. This means that ethylene diamine has two donor atoms. It means that it can bind with the central metal ion with two nitrogen atoms. The bidentate ligands are also known as chelating ligands.
What is difference between denticity and Hapticity?
The key difference between hapticity and denticity is that hapticity refers to the coordination of a ligand to a metal centre via a series of contiguous atoms, whereas denticity refers to the binding of a ligand to a metal centre via covalent chemical bond formation.
How do you identify denticity?
Denticity refers to the number of donor groups in a single ligand that bind to a central atom in a coordination complex. In many cases, only one atom in the ligand binds to the metal, so the denticity equals one, and the ligand is said to be monodentate (sometimes called unidentate).
Is ethylenediamine a bidentate ligand?
Ethylenediamine is a well-known bidentate chelating ligand for coordination compounds, with the two nitrogen atoms donating their lone pairs of electrons when ethylenediamine acts as a ligand.
What is ethylenediamine dihydrochloride?
Ethylenediamine dihydrochloride is a substance that is used to manufacture various drugs and industrial compounds. The most common source of allergic reaction to ethylenediamine dihydrochloride is contact with topical antifungal, antibacterial, and cortisone skin cream mixtures, especially those containing nystatin.
How is ethylenediamine made?
It can be produced in the lab by the reaction of ethylene glycol and urea. Ethylenediamine can be purified by treatment with sodium hydroxide to remove water followed by distillation.
What is hapticity explain with example?
Hapticity is the coordination of a ligand to a metal center via an uninterrupted and contiguous series of atoms. For example, η2 describes a ligand that coordinates through 2 contiguous atoms. In general the η-notation only applies when multiple atoms are coordinated (otherwise the κ-notation is used).