What is titration in chemistry definition?
titration, process of chemical analysis in which the quantity of some constituent of a sample is determined by adding to the measured sample an exactly known quantity of another substance with which the desired constituent reacts in a definite, known proportion.
What is titration in a simple explanation?
Definition of titration : a method or process of determining the concentration of a dissolved substance in terms of the smallest amount of reagent of known concentration required to bring about a given effect in reaction with a known volume of the test solution.
Why is titration important in chemistry?
Titration is important in chemistry as it allows for an accurate determination of solution concentrations of the analyte.
What is titration and its purpose?
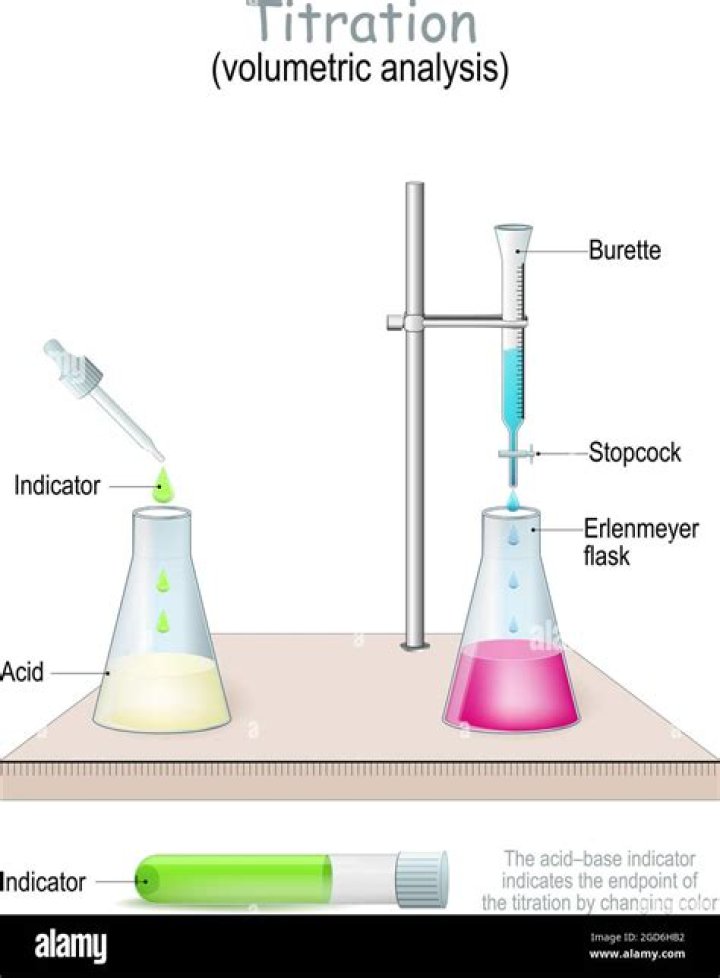
The concentration of a basic solution can be determined by titrating it with a volume of a standard acid solution (of known concentration) required to neutralize it. The purpose of the titration is the detection of the equivalence point, the point at which chemically equivalent amounts of the reactants have been mixed.
What is titration in chemistry class 11?
Titration is the measurement of the volume of a solution of one reactant that is required to react completely with a measured amount of another reactant.
What is titration in chemistry class 12?
Titration is the process or a technique to determine the concentration of an unknown solution, with the use of a solution of known concentration. The concentration of an unknown solution is determined only by knowing the volume of titrant used.
What is the main purpose of titration?
Why is titration performed?
The most common use of titrations is for determining the unknown concentration of a component (the analyte) in a solution by reacting it with a solution of another compound (the titrant). During the course of the titration, the titrant (NaOH) is added slowly to the unknown solution. …
What is titration Ncert?
The process of adding the standard solution to the solution of unknown concentration until the reaction is just complete is called titration. At the completion of the reaction, the indicator shows a visible change e.g. (colour change or turbidity) in the solution being titrated.