What is the Vsepr shape of NBr3?
What is the Vsepr shape of NBr3?
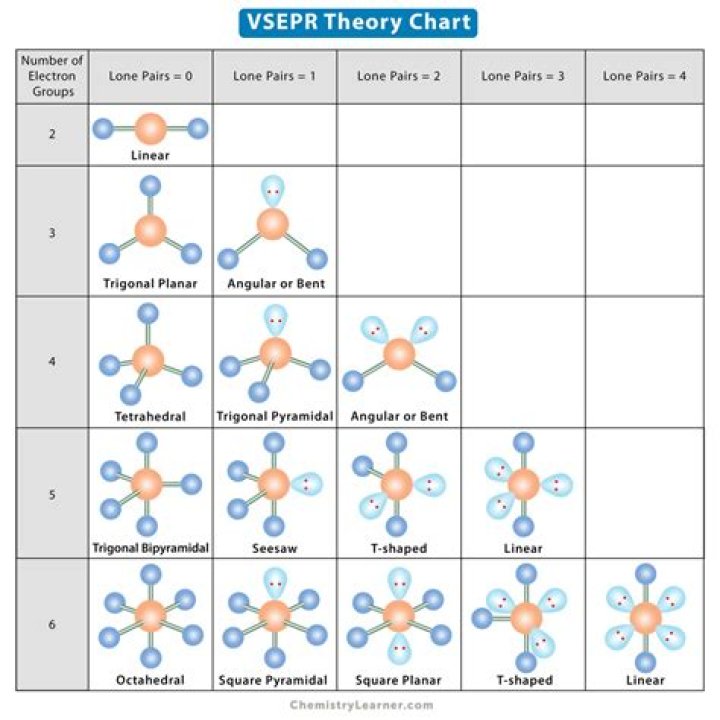
As NBr3 contains one lone pair and three bonded pairs. So, according to the VSEPR chart, the electron geometry of NBr3 is tetrahedral while its molecular geometry is trigonal pyramidal.
What is the correct structure for NBr3?
NBr3Nitrogen tribromide / Formula
How do I find my Vsepr number?
- VSEPR Rules:
- Identify the central atom.
- Count its valence electrons.
- Add one electron for each bonding atom.
- Add or subtract electrons for charge (see Top Tip)
- Divide the total of these by 2 to find the total.
- number of electron pairs.
- Use this number to predict the shape.
Is NBr3 a trigonal pyramidal?
NBr3 is trigonal pyramidal. Molecules with trigonal pyramidal geometry have three atoms at the base of the pyramid and one atom at the apex.
Is NBr3 a polar or nonpolar molecule?
NBr3 has a trigonal pyramidal geometry where the nitrogen atom is bonded to three bromine atoms and has one lone pairs. The individual bonds are polar, as bromine is less electronegative than nitrogen. The overall compound is also polar, as the dipoles do not cancel.
Is NBr3 ionic or covalent?
The bonds in NBr3 N B r 3 are covalent bonds. We have here two nonmetal atoms, and therefore the bonds are going to be covalent.
Is NBr3 a polar molecule?
Is NBr3 polar or nonpolar?
How many lone pairs does sulfur have?
three lone pairs
The sulfur atom shares a bonding pair and three lone pairs. In total, it has six valence electrons.
How do you identify a lone pair and bond pair?
Find the number of lone pairs on the central atom by subtracting the number of valence electrons on bonded atoms (Step 2) from the total number of valence electrons (Step 1). Divide the number of VEs not in bonds (from Step 3) by 2 to find the number of LPs.