What is the valence electrons for carbon?
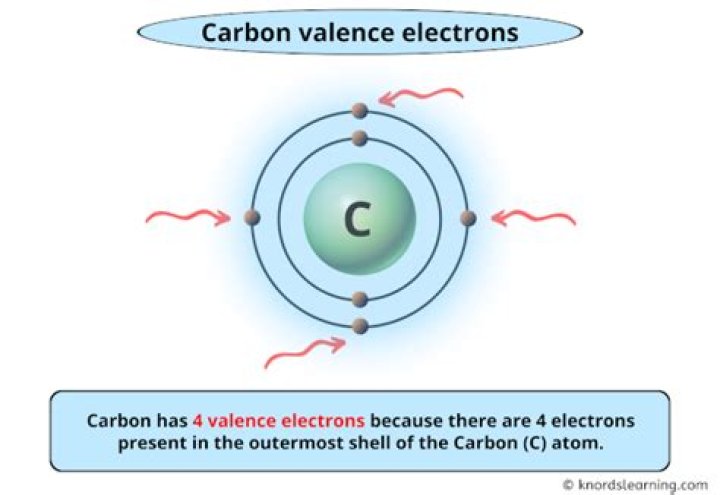
What is the valence electrons for carbon?
Valence electron: An electron in an atom’s valence shell; a lone pair electron or an electron which is part of a covalent bond. Atomic carbon has six electrons: two inner shell (core) electrons in the 1s orbital, and four valence (outer most shell) electrons in the 2s and 2p orbitals.
What is true about the valence electrons?
Valence electrons are the electrons in the outer energy level of an atom that can participate in interactions with other atoms. Valence electrons are generally the electrons that are farthest from the nucleus.
What does carbon do with its valence electrons?
Carbon (4 electrons in the valence shell) combines with four hydrogen atoms to form a stable covalent compound where it shares 8 electrons, while each hydrogen shares 2. Thus every atom in this stable molecule fulfills the octet rule.
How many protons and valence electrons are in carbon?
That means a carbon atom has 6 protons, 6 neutrons, and 6 electrons. Since carbon is in the second row (or second period), it has 2 electron orbits. Use the clay to make your protons and neutrons in the nucleus.
Is it true that the valence electrons are those electrons closest to the nucleus?
Electrons exist in orbitals around a nucleus. Valence electrons are the electrons orbiting the nucleus in the outermost atomic shell of an atom. Electrons that are closer to the nucleus are in filled orbitals and are called core electrons.
Which of the following best explains what a valence electron is?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom.
Why does carbon have 4 valence electrons and not 2?
Explanation: The octet rule states that atoms can fill their outer shells with up to 8 electrons (a full shell of 8 is the most stable configuration). Since Carbon only has 4 of its outer electron slots (or valence electrons) full, it has room to make bonds with 4 other atoms, assuming they are all single bonds.
What is the significance of carbon having four valence electrons?
Two are in a completed inner orbit, while the other four are valence electrons—outer electrons that are available for forming bonds with other atoms. The carbon atom’s four valence electrons can be shared by other atoms that have electrons to share, thus forming covalent (shared-electron) bonds.
Does carbon have 2 or 4 valence electrons?
Explanation: Carbon has six electrons in its neutral state. Valance electrons are the electrons in the outermost energy level. The first two electrons go into the 1 s orbital the lowest energy state.



