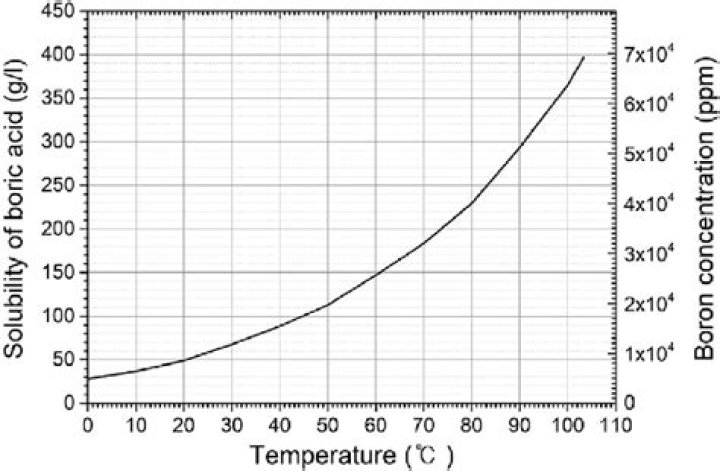
What is the solubility of boric acid?
Water
Alcohol
Boric acid/Soluble in
Boric acid, H3BO3, is moderately soluble in water (roughly 0.4 M at 0 ºC, 0.9 M at 25 ºC and 3 M at 80 ºC). Due to its negative heat of solution, the solubility of boric acid in water increases with temperature.
In which solvent the solubility of boric acid is higher?
Boric acid
| Names | |
|---|---|
| Solubility in water | 2.52 g/100 mL (0 °C) 4.72 g/100 mL (20 °C) 5.7 g/100 mL (25 °C) 19.10 g/100 mL (80 °C) 27.53 g/100 mL (100 °C) |
| Solubility in other solvents | Soluble in lower alcohols moderately soluble in pyridine very slightly soluble in acetone |
| log P | -0.29 |
| Acidity (pKa) | 9.24, 12.4, 13.3 |
How do you increase the solubility of boric acid?
Another method might be to heat the solution to 50-80 C where boric acid is much more soluble. After cooling the supersaturated solution seems stable for some time. Increased pH can give a stable solution with higher B concentration.
How do you dissolve boric acid powder?
Step 2: Dissolve boric acid powder in warm water to make your own weedkiller spray. Put on protective gloves. Mix 10 ounces of boric acid powder into 1/2 cup of warm water, and stir the mixture until all the powder is dissolved.
What pH is boric acid?
5.1
Boric acid/PH
How does boronic acid dissolve?
Solubility / Solution Stability Boric acid dissolves in water: 1 gram in 18 mL cold water, in 4 mL boiling water, 6 mL boiling alcohol.
What can dissolve boric acid?
Solubility / Solution Stability Boric acid dissolves in water: 1 gram in 18 mL cold water, in 4 mL boiling water, 6 mL boiling alcohol.
How strong is boric acid?
Boric acid is a very weak acid with a pKa value of 9.2. At a lower pH than 7, boron is present in its nondissociated form (boric acid) and at a pH greater than 10.5, it is present in the dissociated borate form.
What is the use of boric powder?
It is usually used in dilute solutions as a treatment for diaper rash, insect bites and stings, and sunburns. Boric acid is an effective pesticide for cockroaches, rats, and flies.
Is boronic acid water soluble?
Boronic acids are mild Lewis acids which are generally stable and easy to handle, making them important to organic synthesis….Phenylboronic acid.
| Names | |
|---|---|
| Melting point | 216 °C (421 °F; 489 K) |
| Solubility in water | 10 g/L (20 ºC) |
| Solubility | soluble in diethyl ether, ethanol |
| Acidity (pKa) | 8.83 |