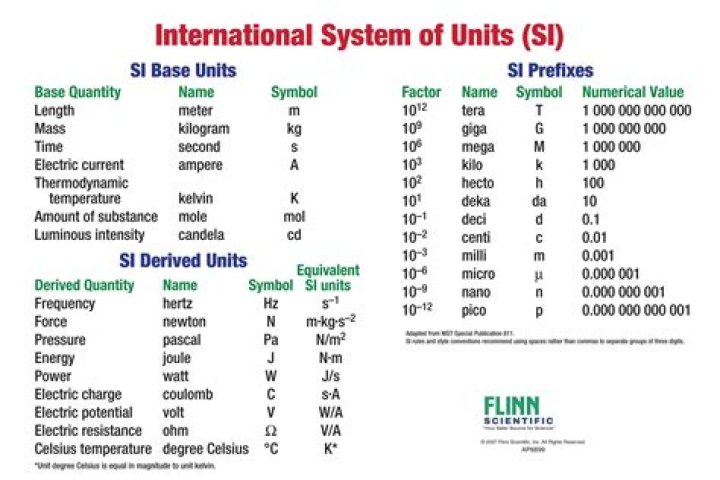
What is the SI unit for measurement of number of particles in a substance group of answer choices?
mole
The mole, symbol mol, is the SI unit of amount of substance. One mole contains exactly 6.022 140 76 x 1023 elementary entities.
What is the SI unit for measurement of number of particles in a substance quizlet?
Chemistry Unit:5 Lesson:7 “The Mole: A Measurement of Matter” What SI unit is used to measure the number of representative particles in a substance? How many hydrogen atoms are in 5 molecules of isopropyl alcohol, C3H7O? You just studied 5 terms!
What is the SI units of amount of a substance?
The SI unit of concentration (of amount of substance) is the mole per cubic meter (mol/m3).
What is the SI unit of the resistance?
ohm
The SI derived unit of electric resistance is the ohm, which is defined as a volt per ampere.
What type of representative particle is NaCl?
The representative particle of an ionic compound is the formula unit. Therefore one mole of sodium chloride contains 6.02 x 1023 formula units of NaCl. The representative particle of a molecular compound is the molecule.
Which of the following is a representative particle?
Molecules. The molecule is the representative particle of molecular compounds. It is also the representative particle of diatomic elements.
How many grams is 5.16 mol Br2?
›› More information from the unit converter You can view more details on each measurement unit: molecular weight of Br2 or grams The SI base unit for amount of substance is the mole. 1 mole is equal to 1 moles Br2, or 159.808 grams.
How do you calculate representative particles?
Multiply the value obtained in Step 3 by Avogadro’s number, which represents the number of representative particles in a mole. Avogadro’s number has a value of 6.02 x 10^23. Continuing the example, 2 moles of water x 6.02 x 10^23 particles per mole = 1.20 x 10^24 particles.
What are the two representative particles?
A representative particle is the smallest unit in which a substance naturally exists. For the majority of elements, the representative particle is the atom. Iron, carbon, and helium consist of iron atoms, carbon atoms, and helium atoms, respectively.
What is the SI unit used to measure amount of substance?
1 mol of a substance is the amount of that substance which contains the same number of particles as 12 g of carbon −12. For solids, the number of moles can found using the following formula: For liquids, the number of moles can found using the following formula: moles = conc.×vol. For gases, 1 mol occupies 24 dm3 at 278 K and 1 atm of pressure.
Which is base unit used to measure the amount of a substance?
the SI base unit used to measure the amount of a substance whose number of particles is the same as the number of atoms of carbon in exactly 12 g of carbon-12. Avogrado’s Number.
What is the SI base unit in Chapter 10?
Chapter 10. the SI base unit used to measure the amount of a substance whose number of particles is the same as the number of atoms of carbon in exactly 12 g of carbon-12.
Can a SI unit be expressed as a multiple?
The SI units can be expressed either as standard multiple or as fractional quantities. These quantities are defined with the help of prefix multipliers with powers of 10 that range from 10 -24 to 10 24. What is the SI Unit?