What is the shape of cl2o?
−
| Central atom: | Cl |
|---|---|
| Total VSEP: | 6 |
| 2 x double bonds: | − 2 pairs |
| Revised Total: | 4 |
| Geometry: | V-shaped (based on tetrahedral) |
What is the compound for cl2o?
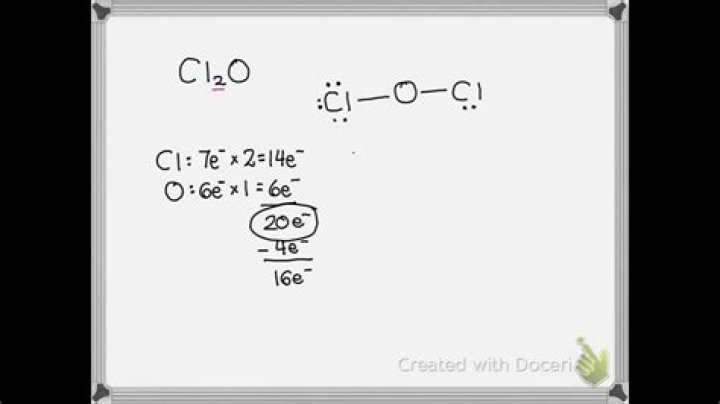
Cl2O
Dichlorine monoxide/Formula
How many valence electrons does cl2o have?
There are a total of 20 valence electrons for the Cl2O Lewis structure. If you check the formal charges for Cl2O you’ll find that the Lewis structure with charges of zero has an Oxygen atom between the two Chlorine atoms.
What’s the structure of Cl2O Is it polar or nonpolar Weegy?
In Cl2O,the polar bonds are arranged asymmetrically around the central atom in a bent shape as there are two lone pair of electrons on the central atom. The bond dipoles do not cancel and the molecule is polar.
What is the central atom in Cl2O?
Lewis structure of Dichlorine monoxide (Cl2O) contains two Cl-O bonds. Oxygen atom is the center atom and both chlorine atoms are located around that center oxygen atom.
What’s the structure of Cl2O Is it polar or nonpolar?
In Cl2O, the polar bonds are arranged asymmetrically around the central atom in a bent shape as there are two lone pair of electrons on the central atom. The bond dipoles do not cancel and the molecule is polar.
How do you form Cl2O?
Dichlorine monoxide is formed by passing chlorine diluted with an inert gas over the surface of a slowly stirred 50% NaOH solution at about 5 °C. The exit gas also contains some unreacted chlorine as well as HOCl and small amounts of water vapor.
What’s the structure of Cl2O Is it polar or non-polar?
What is the oxidation number of Cl2O?
what is oxidation state of chlorine in Cl2O?? as we know oxidation state of oxygen is -2 therefore the equation can be 2x+(-2)=0 where x is the oxidation state of chlorine ..now on solving we get answer as 1.