What is the properties of 2-methylpropan-2-ol?
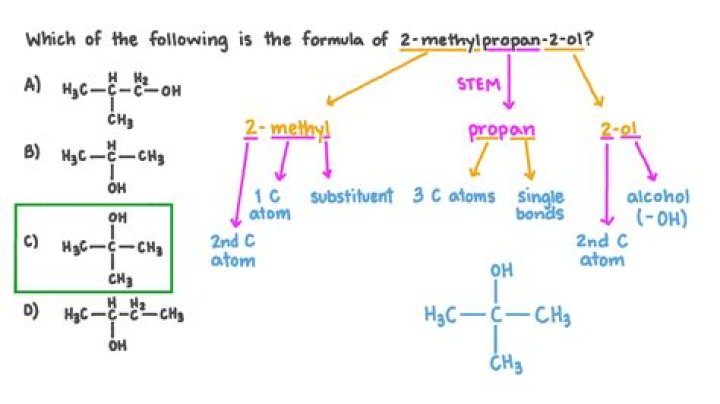
What is the properties of 2-methylpropan-2-ol?
It is one of the four isomers of butanol. It is unique among the isomers of butanol because it tends to be a solid at room temperature, with a melting point slightly above 25 °C. It has a camphor-like odor and is miscible with water, ethanol and diethyl ether.
What is the formula for 2-methylpropan-2-ol?
C₄H₁₀Otert-Butyl alcohol / Formula
Which of the following is the common name for 2-methylpropan-2-ol?
tert-Butanol
| IUPAC Name | 2-methylpropan-2-ol |
|---|---|
| Alternative Names | tert-Butanol tert-Butyl alcohol 2-Methyl-2-propanol 2-Methylpropan-2-ol Trimethylcarbinol t-Butanol |
| Molecular Formula | C4H10O |
| Molar Mass | 74.123 g/mol |
| InChI | InChI=1S/C4H10O/c1-4(2,3)5/h5H,1-3H3 |
Is 2-methylpropan-2-ol a secondary alcohol?
Primary, secondary, and tertiary alcohols The simplest secondary alcohol is isopropanol (propan-2-ol), and a simple tertiary alcohol is tert-butanol (2-methylpropan-2-ol).
Is 2 Methylbutan 2 OL a primary alcohol?
2-methylbutan-2-ol is a tertiary alcohol that is propan-1-ol in which both of the hydrogens at position 1 have been replaced by methyl groups. It has a role as a protic solvent. It is a tertiary alcohol and an aliphatic alcohol.
What type of alcohol is 2-Methylbutan-2-OL?
tertiary alcohol
2-methylbutan-2-ol is a tertiary alcohol that is propan-1-ol in which both of the hydrogens at position 1 have been replaced by methyl groups. It has a role as a protic solvent. It is a tertiary alcohol and an aliphatic alcohol.
How do you oxidize 2-butanol?
Butanol is oxidised by sodium dichromate (Na2Cr2O7) acidified in dilute sulphuric acid to form the aldehyde butanal. The oxidation of the alcohol to an aldehyde is indicated by the colour change of the dichromate solution as it is reduced from the orange colour of Cr2O72− to the green of chromium(III) ions (Cr3+).
What happens when 2-butanol is oxidized?
(a) 2-butanol is a secondary alcohol. So, here the oxidation of the hydroxyl group forms the ketone carboxyl group. Here 2-butanol is oxidized to butanone.
Is 2-Methylbutan-2-OL a primary alcohol?