What is the pOH if the pH is 4?
The pOH Concept Consider a solution with a pH = 4.0. The [H+] of the solution would be 1.0 × 10-4 M. Dividing Kw by this yields a [OH−] of 1.0 × 10-10 M. Finally the pOH of the solution equals -log(1.0 × 10-10) = 10.
How do you find the pOH of a solution?
As with the hydrogen-ion concentration, the concentration of the hydroxide ion can be expressed logarithmically by the pOH. The pOH of a solution is the negative logarithm of the hydroxide-ion concentration. by this yields a [OH − ] of 1.0 × 10 -10 M. Finally the pOH of the solution equals -log(1.0 × 10 -10 ) = 10.
What is the pOH if the pH is 3?
So, if the pH of your acidic solution is three, you can plug this in the equation above to find the pOH: Thus, For a 0.001 M solution of HCl, the pH is three, and the pOH is 11.
What is pOH formula?
To calculate the pOH of a solution you need to know the concentration of the hydroxide ion in moles per liter (molarity). The pOH is then calculated using the expression: pOH = – log [OH-] Example: What is the pOH of a solution that has a hydroxide ion concentration of 4.82 x 10-5 M?
What is pH and pOH scale?
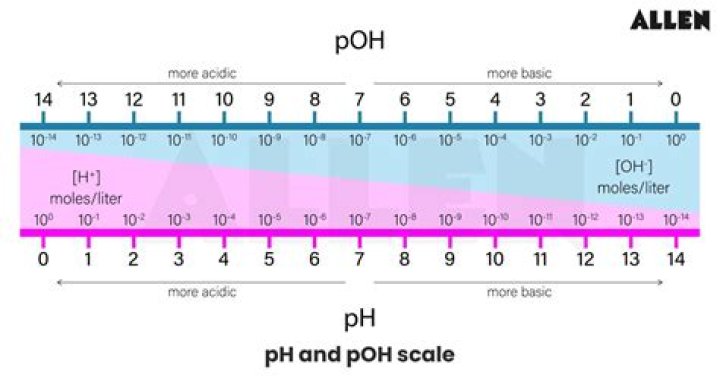
pH and pOH denote the negative log of the concentration of hydrogen or hydroxide ions. High pH means that a solution is basic while high pOH means that a solution is acidic. So here the basic definition of pH is that it’s equal to negative log base 10 of the concentration of protons in your solution.
What is the relation between pH and pOH?
The pOH scale is similar to the pH scale in that a pOH of 7 is indicative of a neutral solution. A basic solution has a pOH less than 7, while an acidic solution has a pOH of greater than 7. The pOH is convenient to use when finding the hydroxide ion concentration from a solution with a known pH.
How is pH and pOH related?
The pH of a solution can be related to the pOH. The pOH scale is similar to the pH scale in that a pOH of 7 is indicative of a neutral solution. A basic solution has a pOH less than 7, while an acidic solution has a pOH of greater than 7.
How is pH calculated?
To calculate the pH of an aqueous solution you need to know the concentration of the hydronium ion in moles per liter (molarity). The pH is then calculated using the expression: pH = – log [H3O+]. On a calculator, calculate 10-8.34, or “inverse” log ( – 8.34).
What is the pOH if the pH is 11?
So, if the pH of your acidic solution is three, you can plug this in the equation above to find the pOH: Thus, For a 0.001 M solution of HCl, the pH is three, and the pOH is 11. You can also do the inverse calculation.
What is the pH of pOH?
High pH means that a solution is basic while high pOH means that a solution is acidic. Neutral solutions have pH and pOH of 7.
What is the formula for phandpoh and Poh?
pH + pOH = 14 If either the pH or the pOH of a solution is known, the other can be
What do you need to know about Poh and pH?
Before you get to the formulas, its important to know more about pH and pOH. You should already know that [H+] is the concentration of hydrogen ions (indicated by the brackets) and that [OH-] is the concentration of hydroxide ions (again, indicated by the brackets).
How to calculate the hydroxide ion concentration ( Poh )?
There are a few different formulas you can use to calculate pOH, the hydroxide ion concentration, or the pH (if you know pOH): pOH = -log 10 [OH – ] [OH – ] = 10 -pOH
How to calculate pH, pOH, H + acids and bases?
Pick one of the formulas: in this case, we are finding
What is the equation which relates pH to Poh?
The chemical equation that ultimately relates the pH of a solution to the pOH at temperatures that are at 25 degrees Celsius would be pH + pOH = pKW which would be 14 at 25 C pH + pOH = 14.
How do you find Poh given pH?
The pH of a solution also can be determined by finding the pOH. Determine the concentration of the hydroxide ions by dividing the molecules of hydroxide by the volume of the solution. Take the negative log of the concentration to get the pOH. Then subtract this number from 14 to get the pH.
How do I find Poh from pH?
So, if you know the hydroxide concentration, you can calculate the pH with the equation 14 – pOH = pH. For example, if a substance has a pOH of 12, it would have a pH of 2. You can apply the same principal in reverse to find pOH from pH, using the formula 14 – pH = pOH.
How do you calculate Poh?
Calculating pOH. The calculation for pOH follows the same rules as for pH, but uses the concentration of OH – ions: pOH = – log(OH -). For example, find the pOH of a 0.02 M solution of sodium hydroxide (KOH).