What is the pKa of acetic acid?
What is the pKa of acetic acid?
4.75
Ionization Constants of Heteroatom Organic Acids
| Common Name | Formula | pKa |
|---|---|---|
| formic acid | HCO2H | 3.75 |
| ascorbic acid | 4.17 11.6 | |
| benzoic acid | C6H5CO2H | 4.20 |
| acetic acid | CH3CO2H | 4.75 |
Can the pKa of acetic acid be calculated?
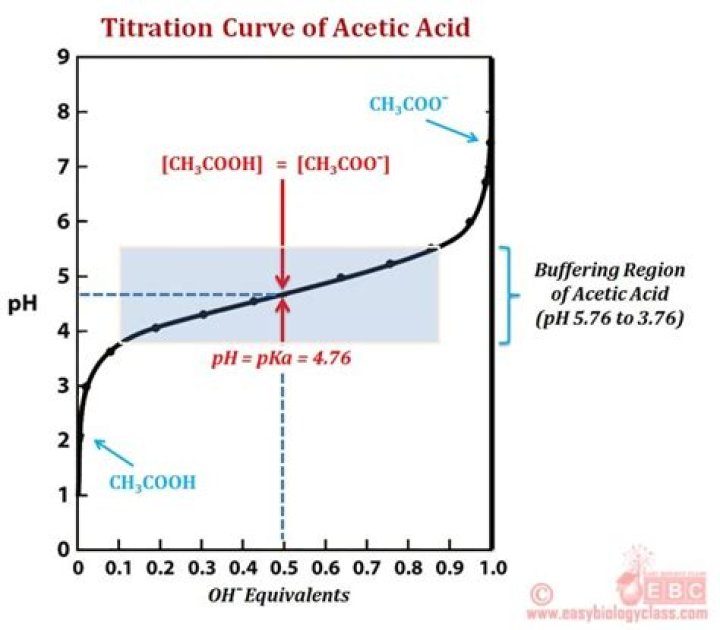
The pKa of acetic acid is 4.76, which is the pH value at which half of the acid will be intact and the other half in the ionic form. The formula for the acetate ion (the conjugate base of acetic acid) is CH3COO–.
What is the pKa of ch3nh?
For instance, here’s a pKa table where the pKa of methylamine (CH3NH2) is listed as 10.63.
What is the pKa of c6h5o?
–) react with phenol (C6H5OH)? Data: H2CO3 (pKa = 6.35), C6H5OH (pKa = 10.0).
What is the N factor of acetic acid?
CH3COOH has n factor of 1.
What is pKb?
pKb is the negative base-10 logarithm of the base dissociation constant (Kb) of a solution. It is used to determine the strength of a base or alkaline solution.
How do you calculate pKa from pH?
Each dissociation has a unique Ka and pKa value. When the moles of base added equals half the total moles of acid, the weak acid and its conjugate base are in equal amounts. The ratio of CB / WA = 1 and according to the HH equation, pH = pKa + log(1) or pH = pKa.
What is the pKa of CH3CH2NH2?
The value of pKb for the base ethylamine (CH3CH2NH2 C H 3 C H 2 N H 2 ) is 3.19 at 25C, while the pKa for formic acid (HCO2H H C O 2 H ) is 3.74, also at 25C.
What is the pKa of NH4+?
9.26
pKa of NH4^ + is 9.26 .
How do you find n Factor?
To calculate n-factor of a salt of such type, we take one mole of the reactant and find the number of mole of the element whose oxidation state is changing. This is multiplied with the oxidation state of the element in the reactant, which gives us the total oxidation state of the element in the reactant.



