What is the meaning of atomic model?
2. any entity regarded as the indivisible building block of a theory. 3. ( Philosophy) the hypothetical indivisible particle of matter postulated by certain ancient philosophers as the fundamental constituent of matter.
What is the atomic model called?
Rutherford model, also called Rutherford atomic model, nuclear atom, or planetary model of the atom, description of the structure of atoms proposed (1911) by the New Zealand-born physicist Ernest Rutherford.
What is the simple atomic model?
The nucleus of an atom contains protons and neutrons. The nucleus carries a positive electrical charge. Electrons move around outside the nucleus. Simple models show electrons orbiting the nuclear in a near-circular orbit, like planets orbiting a star, but real behavior is much more complex.
Why is it called atomic model?
Atomic theory traces its origins to an ancient philosophical tradition known as atomism. Ancient Greek philosophers called these hypothetical ultimate particles of matter atomos, a word which meant “uncut”.
What is atomic model in physics?
Rutherford’s model of the atom Nucleus contains relatively high central charge concentrated into very small volume. This small volume also contains the bulk of the atomic mass of the atom. Nucleus is surrounded by lighter and negatively charged electrons. His model is sometimes known as the planetary model of the atom.
What is the function of an atomic model?
Complex orbitals
| ℓ = 0 (s) | ℓ = 1 (p) | |
|---|---|---|
| n = 1 | ||
| n = 2 | 0 | −1, 0, 1 |
| n = 3 | 0 | −1, 0, 1 |
| n = 4 | 0 | −1, 0, 1 |
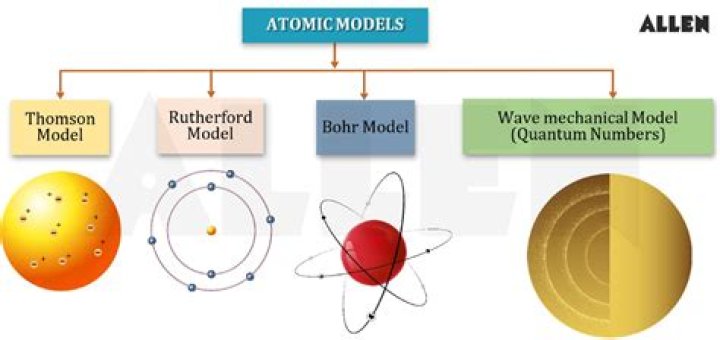
What are all the atomic models?
Thomson’s model: Plum Pudding model. Ernest Rutherford’s model: Nuclear model. Niels Bohr’s model: Planetary model. Erwin Schrödinger’s model: Electron Cloud Model/Quantum Model.
Why is the atomic model important?
Atomic models are important because, they help us visualize the interior of atoms and molecules, and thereby predicting properties of matter.
What is called atom?
atom, smallest unit into which matter can be divided without the release of electrically charged particles. It also is the smallest unit of matter that has the characteristic properties of a chemical element.