What is the magnetic quantum number for P orbital?
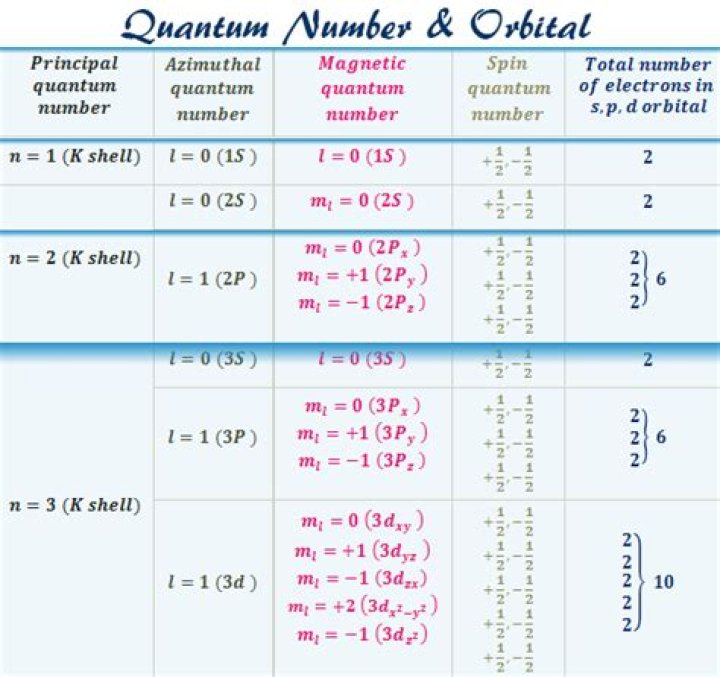
Quantum Numbers and Atomic Orbitals
| l | 0 | 4 |
|---|---|---|
| Letter | s | g |
What is the symbol for magnetic quantum number?
symbol ml
The magnetic quantum number (symbol ml) is one of four quantum numbers in atomic physics.
What does the quantum number P represent?
The Magnetic Quantum Number The p subshell (ℓ = 1) contains three orbitals (in some systems depicted as three “dumbbell-shaped” clouds), so the mℓ of an electron in a p subshell will be −1, 0, or 1.
What is the magnetic quantum number for 3p?
3
CORRECT: For the 3p sublevel, the principal quantum number (n) is 3 and the angular momentum quantum number (l) is 1.
What is the value of M for DXY orbital?
Once principle quantum number n equals 3 or greater, angular quantum number can equal 2. When angular quantum number l=2, it is considered the d-orbital. For the d-orbital, the magnetic quantum number ml can equal -2 to 2, taking the possible values -2, -1, 0, 1, or 2.
What is the shape of DXY?
The ml values for five d orbitals are -2, -1, 0, +1, and +2 i.e., we can say d-subshell has five orientations. All these d-orbitals have the same energy and are called degenerate orbitals. The shape of the d-orbitals is given below: Hence, we can say d-orbitals have double dumbbell-shaped.
What is shape of P orbital?
A p orbital has the approximate shape of a pair of lobes on opposite sides of the nucleus, or a somewhat dumbbell shape. An electron in a p orbital has equal probability of being in either half.
What does the magnetic quantum number tell us and what letter is used as its variable?
The magnetic quantum number tells us how many orbitals there are in a subshell and the possible orientations of these orbitals. Its value is dependent on the angular momentum quantum number, l.
What are the values of quantum number n L and M for 3p?
for 3p-orbitals,, n=3, l=1 and m=+1,0,-1. (iiii) n=3, l=1, m=-1.
How many orbitals are in P?
3 orbitals
The p sublevel has 3 orbitals, so can contain 6 electrons max.