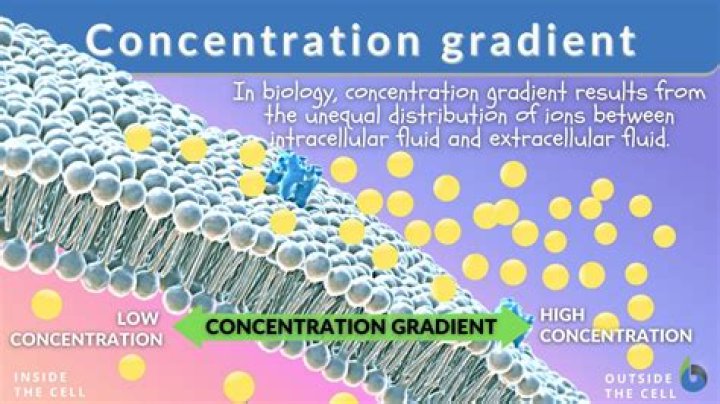
What is the H+ concentration gradient?
When the concentration of something builds up on only one side of a membrane, we call this a concentration gradient. Concentration is high on one side, but low on the other. Let’s think of this in terms of a hydrogen ion, H+. Hydrogen ions naturally move down this concentration gradient, from high to low concentration.
How do you find the OH?
Find the [OH-] given the pH or pOH. You are given that the pH = 4.5. Find the hydroxide ion concentration of a solution with a pOH of 5.90. To solve this, use a scientific calculator and enter 5.90 and use the +/- button to make it negative and then press the 10x key.
Is H+ equal to OH?
In pure water, the hydrogen ion concentration, [H+], equals the hydroxide ion concentration, [OH-]. These concentrations can be calculated from the equation for the ionization of water.
What is the relationship between KA and KB with KW?
What would we do with Ka and Kb to get Kw? It turns out that you multiply them, Ka times Kb for a conjugate acid-base pair is equal to Kw.
How is the concentration gradient of H+ maintained?
as the inner mitochondrial membrane is impermeable to protons, H+ ions cannot pass back through the inner mitochondrial membrane, into the matrix, therefore there is a higher concentration of H+ ions in the inter-membrane space, thus setting up and maintaining a proton gradient across the inner mitochondrial membrane.
How is the H+ gradient used to make ATP?
The proton gradient produced by proton pumping during the electron transport chain is used to synthesize ATP. Protons flow down their concentration gradient into the matrix through the membrane protein ATP synthase, causing it to spin (like a water wheel) and catalyze conversion of ADP to ATP.
Is acid H or OH?
Acids are substances that provide hydrogen ions (H+) and lower pH, whereas bases provide hydroxide ions (OH–) and raise pH. The stronger the acid, the more readily it donates H+. For example, hydrochloric acid and lemon juice are very acidic and readily give up H+ when added to water.
Where do H+ and OH ions come from?
Where do Hydrogen Ions (H+) come from? Well, it’s all part of water really… Water (H2O) splits into Hydrogen Ions (H+) and Hydroxyl Ions (OH-). When there are equal parts of Hydrogen Ions (H+) and Hydroxyl Ions (OH-) leading to a 1:1 ratio, pH is neutral (7).
What happens to OH when H+ increases?
As the concentration of hydrogen ions in a solution increase, the more acidic the solution becomes. As the level of hydroxide ions increases the more basic, or alkaline, the solution becomes. Thus, as the hydrogen ion concentration increases hydroxide ion concentration falls, and vice versa.