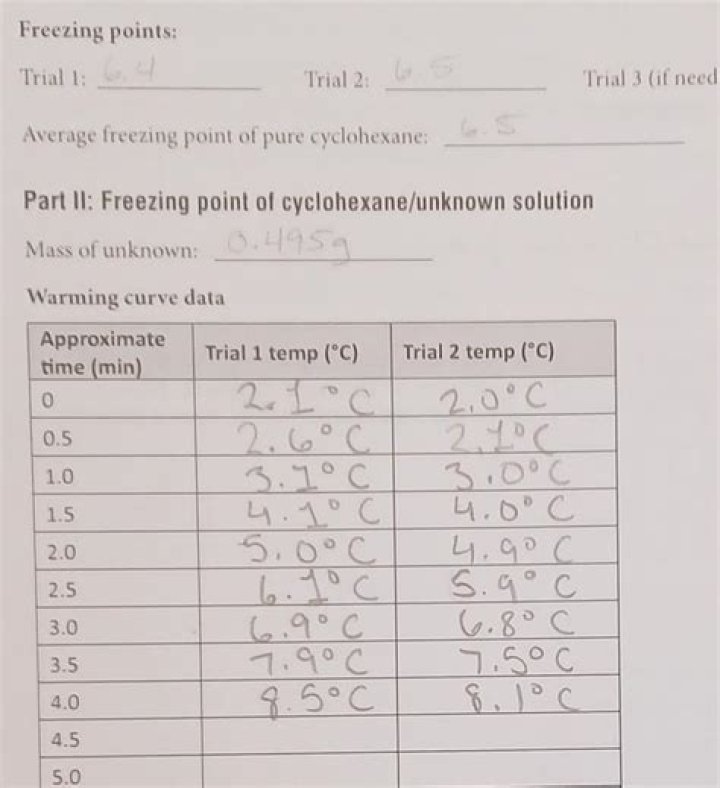
What is the freezing point of cyclohexane?
6.50 °C.
Exercise: The freezing point of cyclohexane is 6.50 °C.
What is the freezing point depression of naphthalene?
Sample calculation. The freezing-point depression constant for naphthalene is 6.90 oC/m.
What is the normal freezing point of naphthalene?
Calculations. Using 80.1 oC as the theoretical value for the freezing point of naphthalene, we can now determine percent error.
What is the freezing point of naphthalene in benzene?
1.60 g of naphthalene (C10H8) is dissolved in 20.0 g of benzene. The freezing point of pure benzene is 5.5 oC, and the freezing point of the mixture is 2.8 oC.
What is the freezing point of butanol?
-129.6°F (-89.8°C)
1-Butanol/Melting point
What’s freezing point?
Medical Definition of freezing point : the temperature at which a liquid solidifies specifically : the temperature at which the liquid and solid states of the substance are in equilibrium at atmospheric pressure : melting point the freezing point of water is 0° Celsius or 32° Fahrenheit.
What freezing point means?
freezing point, temperature at which a liquid becomes a solid. As with the melting point, increased pressure usually raises the freezing point. The freezing point is lower than the melting point in the case of mixtures and for certain organic compounds such as fats.
How do you find freezing point?
The freezing point depression ∆T = KF·m where KF is the molal freezing point depression constant and m is the molality of the solute. Rearrangement gives: mol solute = (m) x (kg solvent) where kg of solvent is the mass of the solvent (lauric acid) in the mixture. This gives the moles of the solute.
What is the freezing point depression constant of T butanol?
Problem: The freezing point of t-butanol is 25.50°C and Kf is 9.1°C • kg/mol. Usually t-butanol absorbs water on exposure to air.