What is the formal charge of n2h2?
3.1Computed Properties
| Property Name | Property Value | Reference |
|---|---|---|
| Heavy Atom Count | 2 | Computed by PubChem |
| Formal Charge | 0 | Computed by PubChem |
| Complexity | 0 | Computed by Cactvs 3.4.8.18 (PubChem release 2021.05.07) |
| Isotope Atom Count | 0 | Computed by PubChem |
What is the formal charge on a carbene?
(One last possibility is a highly reactive species called a ‘carbene’, in which a carbon has two bonds and one lone pair of electrons, giving it a formal charge of zero.
How do you calculate the formal charge of sulfur?
Continuing with sulfur, we observe that in (a) the sulfur atom shares one bonding pair and has three lone pairs and has a total of six valence electrons. The formal charge on the sulfur atom is therefore 6 – (6 + 2/2) = 1−.
How do you calculate the formal charge of nitrogen?
Continuing with the nitrogen, we observe that in (a) the nitrogen atom shares three bonding pairs and has one lone pair and has a total of 5 valence electrons. The formal charge on the nitrogen atom is therefore 5 – (2 + 6/2) = 0.
How do you find the formal charge on a resonance structure?
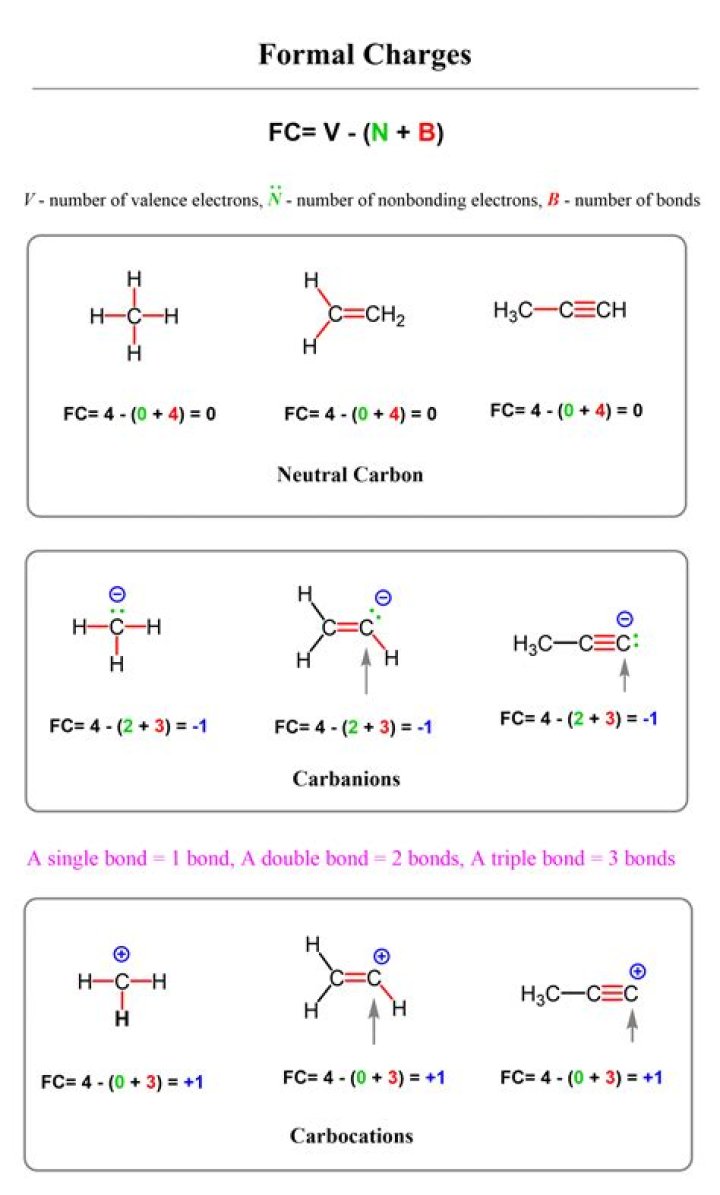
Calculating Formal Charge Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and then subtract the number of bonds connected to that atom in the Lewis structure.
How do you calculate formal charge?
Since a chemical bond has two electrons, the “number of bonding electrons divided by 2” is by definition equal to the number of bonds surrounding the atom. So we can instead use this shortcut formula: Formal Charge = [# of valence electrons on atom] – [non-bonded electrons + number of bonds].
Why do we calculate formal charge?
Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.
Is N2H2 empirical formula?
H2N2Diimide / Formula