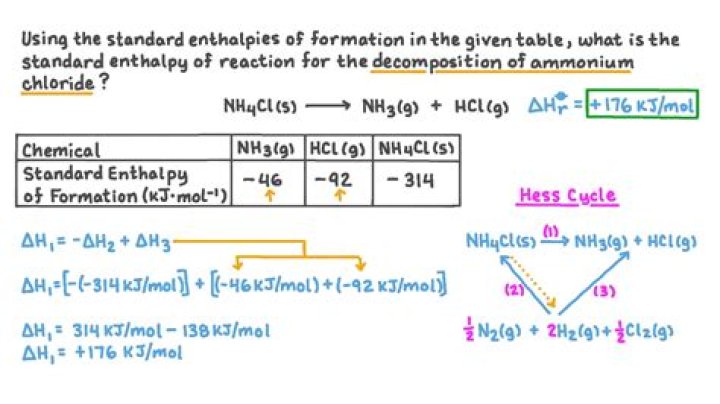
What is the enthalpy of the decomposition of NH4Cl?
The official ΔH dissolution for NH4CL was 14780 J/mol at rtp, the experimental value was 11750 J/mol.
Is the decomposition of NH4Cl exothermic?
The decomposition of ammonium chloride into ammonia and hydrogen chloride is endothermic (heat absorbed or heat taken in from the surroundings – the hot test tube).
What is the enthalpy change for the decomposition of water?
285.8 kJ
Notice that the decomposition of water (equation b) is endothermic and requires the input of 285.8 kJ energy per mole of water decomposed. The reverse reaction, the formation of one mole of water from hydrogen and oxygen (equation d), is exothermic and releases 285.8 kJ energy.
What is the reaction between ammonium chloride and water?
When you dissolve ammonium chloride crystals in water (H2O), the ammonium chloride compound decomposes into its component ions: NH4+ and Cl-. The dissociation chemical reaction is: NH4Cl(solid) = NH4+(aqueous) + Cl-(aqueous).
What is the lattice enthalpy of NH4Cl?
Use the enthalpy changes shown and a Born-Haber type cycle to calculate the enthalpy of solution of ammonium chloride, NH4Cl (s): Lattice energy of NH4Cl (s) = –705 kJ/mol Enthalpy of hydration NH4+ (g) = –307.
Is ammonium chloride and water exothermic or endothermic?
When ammonium chloride dissolves in water, the absorption of heat occurs shows the reaction is an endothermic reaction.
Is NH4Cl h2o endothermic or exothermic?
The same thing happens when ammonium chloride is dissolved in water. The endothermic separation of the ions will dictate the overall reaction, since the exothermic hydration of the ions will gfive off less energy. As a result, dissolving ammonium chloride in water will be an endothermic process.
Why is the decomposition of water endothermic?
The decomposition of water into hydrogen and oxygenWhen water is heated to over 2000 degrees Celsius, a small fraction will decompose into hydrogen and oxygen. Significant heat energy is needed for this reaction to proceed, so the reaction is endothermic.
Is ammonium chloride a sublime?
Ammonium chloride appears to sublime upon heating but actually decomposes into ammonia and hydrogen chloride gas.