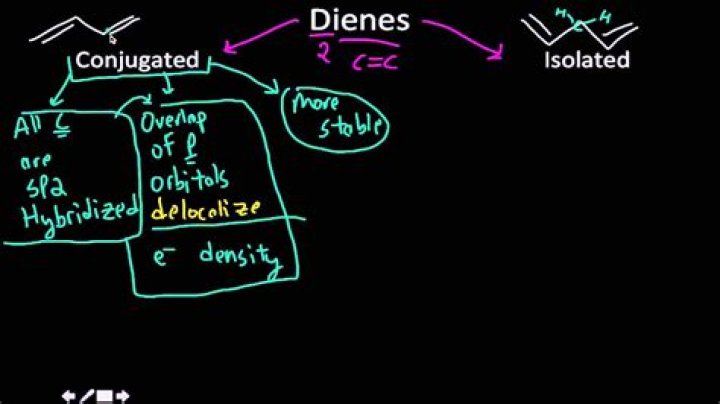
What is the difference between conjugated and isolated dienes?
Conjugated dienes have conjugated double bonds separated by one single bond. Unconjugated dienes have the double bonds separated by two or more single bonds. They are usually less stable than isomeric conjugated dienes. This can also be known as an isolated diene.
What is a isolated alkene?
When the double bonds are separated from each other by two or more single bonds, they are called isolated double bonds. Isolated double bonds undergo normal alkene reactions, revealing that no interaction occurs between them. Such an arrangement is called a conjugated double‐bond system.
Which diene is the most stable?
conjugated dienes
This extra bonding interaction between the adjacent π systems makes the conjugated dienes the most stable type of diene. Conjugated dienes are about 15kJ/mol or 3.6 kcal/mol more stable than simple alkenes.
What are cumulated dienes give examples?
The 1,2-dienes, which have cumulated double bonds, commonly are called allenes. The simplest example is 1,2-propadiene, Figure 13-4). In this formulation each of two electrons of the central atom form collinear spσ bonds to the terminal sp2-hybridized carbons.
Which one is a cumulated diene?
Allylene. H2C=C=CH2 (Allene) Allene is an example of cummulated diene.
What are isolated bonds?
If double bonds are separated by one or more saturated carbon atoms, they are said to be isolated. If they are separated by only one single bond, they are said to be conjugated. Two double bonds on one carbon are said to be cumulated.
What is meant by allylic carbon?
An allylic carbon is a carbon atom bonded to a carbon atom that in turn is doubly bonded to another carbon atom. see also allylic hydrogen.
How do you name diene?
The rule for naming is they all end with “-diene”. Naming Compounds- Again, ignore all the hydrogen’s. We only worry about carbon atoms.
How do you make diene?
Dienes are prepared from the same reactions that form ordinary alkenes. The two most common methods are the dehydration of diols (dihydroxy alkanes) and the dehydrohalogenation of dihalides (dihaloalkanes). The generation of either an isolated or conjugated system depends on the structure of the original reactants.
Which of the following is most stable isolated Diene?
Correct Option- i Conjugated alkadiene CH2 =CH—CH =CH2 Explanation-i Due to resonance conjugated alkadiene is maximum stable. Isolated is more stable than cumulated alkadiene due to H-effect .
How do you check the stability of a dienes?
The correct order of stability of dienes is conjugated > isolated > cumulated.