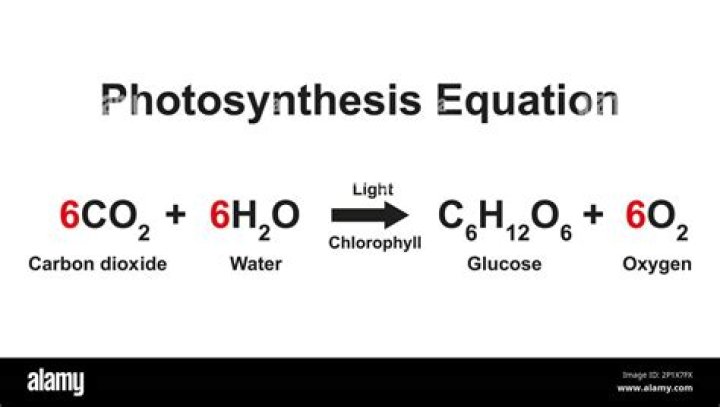
What is the delta G for photosynthesis?
ΔGo’ = +2840 kJ/mol (Unfavorable ΔGo’ explains the need for energy input.) The light reactions occur on the thylakoid membranes of chloroplasts (or the plasma membrane or internal membranes of bacteria).
Is photosynthesis negative or positive delta G?
This is expressed in terms of the free energy ΔG° of the reaction, which for photosynthesis is +479 kJ ⋅ mol-1 or 479 joules of energy per mole. The positive sign indicates an endothermic reaction, while a negative sign indicates an exothermic process.
What is Delta G RXN?
Every chemical reaction involves a change in free energy, called delta G (∆G). The change in free energy can be calculated for any system that undergoes a change, such as a chemical reaction. To calculate ∆G, subtract the amount of energy lost to entropy (denoted as ∆S) from the total energy change of the system.
What is Delta G in respiration?
A reaction actually describes a change (delta) from one state to another. The change in free energy is denoted as delta G. If this value is negative, then free energy will leave the system, work can be done, and the reaction will occur spontaneously. Enthalpy (H) is the total energy in a system.
Is Delta G in photosynthesis positive?
The positive sign of the standard free energy change of the reaction (ΔG°) given above means that the reaction requires energy (an endergonic reaction). The energy required is provided by absorbed solar energy, which is converted into the chemical bond energy of the products (Box 1).
Is photosynthesis Delta G positive?
Introduction. Photosynthesis is the ultimate source of all of humankind’s food and oxygen, whereas fossilized photosynthetic fuels provide ∼87% of the world’s energy. The positive sign of the standard free energy change of the reaction (ΔG°) given above means that the reaction requires energy (an endergonic reaction).
How do you find Delta G RXN?
ΔG=ΔG0+RTlnQ where Q is the ratio of concentrations (or activities) of the products divided by the reactants. Under standard conditions Q=1 and ΔG=ΔG0 . Under equilibrium conditions, Q=K and ΔG=0 so ΔG0=−RTlnK . Then calculate the ΔH and ΔS for the reaction and the rest of the procedure is unchanged.
How do you find Delta S RXN?
Starts here2:4415.2 Calculate the standard entropy change for a reaction [HL IB …YouTube
How is Delta G affected by the enzyme?
Enzymes decrease the Gibbs free energy of activation, but they have no effect on the free energy of reaction. Enzymes work by lowering the activation energy ( Ea or ΔG✳ ) for a reaction. This increases the reaction rate.
What are the Assimilatory power?
Assimilatory power is the power of plants in the form of ATP & NADPH (produce during light reactions) to obtain food in the form of carbohydrates from the reduction of CO2 during photosynthesis.