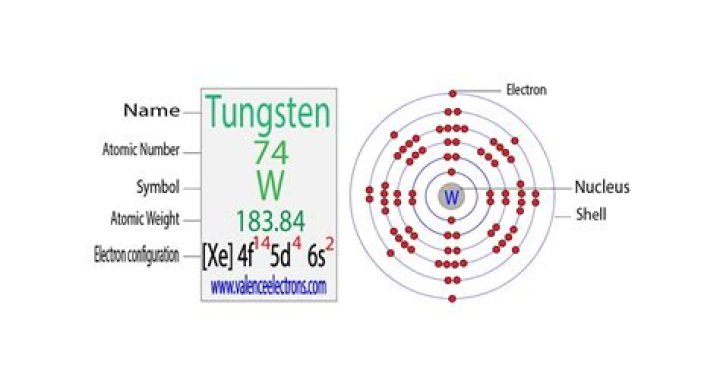
What is the complete electron configuration for tungsten?
[Xe] 6s² 4f¹⁴ 5d⁴Tungsten / Electron configuration
Which atom has the electron configuration 1s2 2s2 2p6 3s2 3p6 4s2 3d2?
If you are referring to a neutral atom, then Vanadium (V) has that particular electron configuration.
Which element has an electron configuration of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d1?
antimony atom
So, an antimony atom with charge +2 has an electron configuration of 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p1.
What element has this configuration 1s2 2s2 2p6 3s2?
2 Answers. BRIAN M. The electron configuration 1s22s22p63s23p2 is the element Silicon.
What is the group number of the element Tungsten?
Group 6
Fact box
| Group | 6 | 3414°C, 6177°F, 3687 K |
|---|---|---|
| Atomic number | 74 | 183.84 |
| State at 20°C | Solid | 182W, 184W, 186W |
| Electron configuration | [Xe] 4f145d46s2 | 7440-33-7 |
| ChemSpider ID | 22403 | ChemSpider is a free chemical structure database |
What is the spin quantum number for Tungsten symbol W )?
What is the spin quantum number for Tungsten (symbol W)? Tungsten has 4 electrons in the 5d orbital. Therefore 1 electron will go into each orbital (no pairing). The 4th electron will have a +1/2 spin.
What element is 1s2 2s2 2p6 3s2 3p6 4s2 3d3?
Vanadium
Example: 1s2 2s2 2p6 3s2 3p6 4s2 3d3 (This has a total of 23 electrons which equals the atomic number of V – Vanadium.)
What element is 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3?
Electron Configuration Matching Game
| A | B |
|---|---|
| 1s2 2s2 | Be |
| 1s2 2s2 2p3 | N |
| 1s2 2s2 2p6 3s2 3p6 4s2 3d3 | V |
| 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p3 | As |