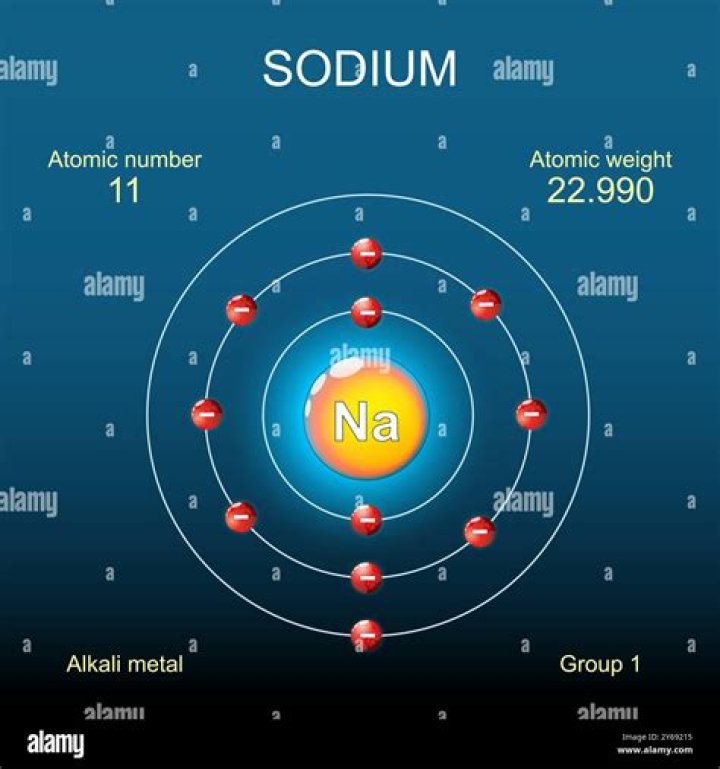
What is the Bohr Rutherford diagram for sodium?
Below is an illustration of the Bohr model of a sodium atom. The electron in the M-shell is weakly attracted to the nucleus. If you look at the diagrams of the sodium and chlorine atoms you can see that sodium normally has eleven electrons in shells around the nucleus.
What does one atom of sodium look like?
The sodium atom has 11 electrons and 11 protons with one valence electron in the outer shell. Sodium in its pure form is very reactive. It is a very soft metal that can be easily cut with a knife. It is silvery-white in color and burns with a yellow flame.
How many electrons are in a shell?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.
What is the correct Bohr electron configuration for the sodium ion Na +?
The electron configuration of a neutral sodium atom is 1s22s22p63s1 . In this configuration we note that there is only one electron in the 3rd energy level. Atoms prefer to gain the stability of octet, by having eight electrons in the outer shell, the electrons of the s and p orbitals.
What is the ground state electron configuration of the sodium ion Na+?
Examples: Na has a ground-state electronic configuration of 1s2 2s2 2p6 3s1. Removing the 3s electron leaves us with the noble gas configuration 1s2 2s2 2p6, so a sodium ion is Na+.
What are the 5 parts of an atom?
What Are The 5 Parts Of An Atom
- Protons.
- Neutrons.
- electrons.
Why is an atom neutral?
Electrons have electric charge of -1 and the number of electrons in an atom is equal to the number of protons. Heavier atoms tend to have more neutrons than protons, but the number of electrons in an atom is always equal to the number of protons. So an atom as a whole is electrically neutral.
What’s the charge of a sodium atom?
+1
A sodium atom can lose its outer electron. It will still have 11 positive protons but only 10 negative electrons. So, the overall charge is +1. A positive sign is added to the symbol for sodium, Na +.What is the structure of the sodium atom?
The nucleus consists of 11 protons (red) and 12 neutrons (blue). 11 electrons (green) bind to the nucleus, with a single, relatively unstable electron in the outer shell (ring). The stability of an element’s outer electrons determines its chemical and physical properties.
How to draw an atomic structure for sodium?
So, if you want to draw atomic structure for sodium first of all you should know the atomic number for sodium. Atomic number of sodium is 11. Atomic number of any element refers to the number of electrons in an atom of that element they are having. STEP 1. Always write the atomic number of the element first.
How can I make a sodium atom model?
STEP 1. Always write the atomic number of the element first. Once you know its atomic number you know the number of protons and electrons in it. As in every stable atom the number of protons is equal to the number of electrons. Therefore, now you know the number of electrons too. STEP 2. Do the electronic configuration for the atom of the element.
How do you draw a diagram of an atom?
Draw one ring around the atom’s nucleus for each shell the atom has. Draw small circles on each ring to represent the number of electrons on that shell. The first shell is the ring closest to the nucleus.
How to calculate the atomic number of an element?
Atomic number of any element refers to the number of electrons in an atom of that element they are having. STEP 1. Always write the atomic number of the element first. Once you know its atomic number you know the number of protons and electrons in it. As in every stable atom the number of protons is equal to the number of electrons.
How many atoms are in a sodium atom?
So for your question, the Periodic Table tells us that sodium has an Atomic Number of 11, so there are 11 protons and 11 electrons. The Periodic Table tells us that sodium has an Atomic Mass of ≈23. So there are 23 – 11 = 12 neutrons. Click to see full answer
What are the properties of a sodium atom?
The sodium atom has 11 electrons and 11 protons with one valence electron in the outer shell. Characteristics and Properties. Sodium in its pure form is very reactive. It is a very soft metal that can be easily cut with a knife. It is silvery-white in color and burns with a yellow flame.
What does an atom of sodium look like?
Sodium is a chemical element with the symbol Na (from Latin natrium) and atomic number 11. It is a soft, silvery-white, highly reactive metal.
Which statements describe sodium atoms?
Sodium is an atom that has 11 protons and 12 neutrons in its nucleus and 11 electrons circling around its nucleus. Like other light atoms such as carbon, sodium forms inside of stars that are beginning to run out of fuel, and it scatters all over space when that star explodes in a supernova.