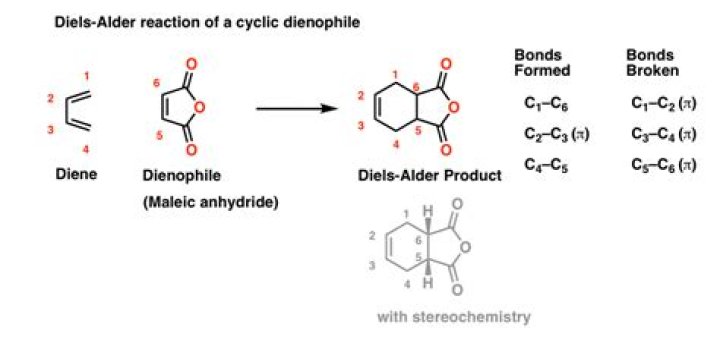
What is the best dienophile for a Diels Alder reaction?
Maleic anhydride is also a very good dienophile, because the electron-withdrawing effect of the carbonyl groups causes the two alkene carbons to be electron-poor, and thus a good target for attack by the pi electrons in the diene.
Is cyclopentadiene a dienophile?
We already know that cyclopentadiene is a good diene because of its inherent s-cis conformation. In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups) and electron-withdrawing groups on the dienophile.
What makes a good dienophile?
Good dienophiles often bear one or two of the following substituents: CHO, COR, COOR, CN, C=C, Ph, or halogen. The diene component should be as electron-rich as possible. There are “inverse demand” Diels Alder Reactions that involve the overlap of the HOMO of the dienophile with the unoccupied MO of the diene.
Which diene reacts more rapidly in Diels Alder reactions cyclopentadiene or 1/3 butadiene briefly explain your choice?
The cyclopentadiene will react more quickly. The reason why the cyclopentadiene reacts more quickly is because it has a fixed conformation….
What is the Diels-Alder product between two molecules of cyclopentadiene?
Cyclopentadiene Slowly Undergoes A Diels-Alder Reaction With Itself To Give “Dicyclopentadiene”, Which Reverts Back To Cyclopentadiene Upon Heating To 180°C.
Which dienophile is the most reactive?
The most reactive dienophile is the aldehyde — propenal.
What diene and dienophile would be the retro Diels-Alder products of?
The retro Diels-Alder reaction (rDA) is the microscopic reverse of the Diels-Alder reaction—the formation of a diene and dienophile from a cyclohexene.
What makes a bad dienophile?
The Ph substituent is not an electron-withdrawing group making this a bad dienophile. The dienophile has a carbonyl but it is not attached directly (i.e. conjugated) to the alkene so cannot activate it. The O atom is deactivating because it is an electron withdrawing group.
Why is cyclopentadiene more reactive than butadiene?
DMCP and cyclopentadiene are more reactive than butadiene because they are locked into s-cis conformation, whereas furan is less reactive than the other dienes because it is somewhat aromatic. Intramolecular Reactivity of Cyclic Dienes with Cyclobutadiene.
Which Diels Alder reaction is faster?
In general, Diels-Alder reactions proceed fastest with electron-donating groups on the diene (eg. alkyl groups) and electron-withdrawing groups on the dienophile.
Why is cyclopentadiene reactive?
Cyclopentadiene is highly reactive in Diels–Alder reactions because only minimal out-of-plane distortion is required to achieve the transition state geometry compared with that of other cyclic and acyclic dienes. Asynchronous transition states have significant out-of-plane distortion about only one double bond.