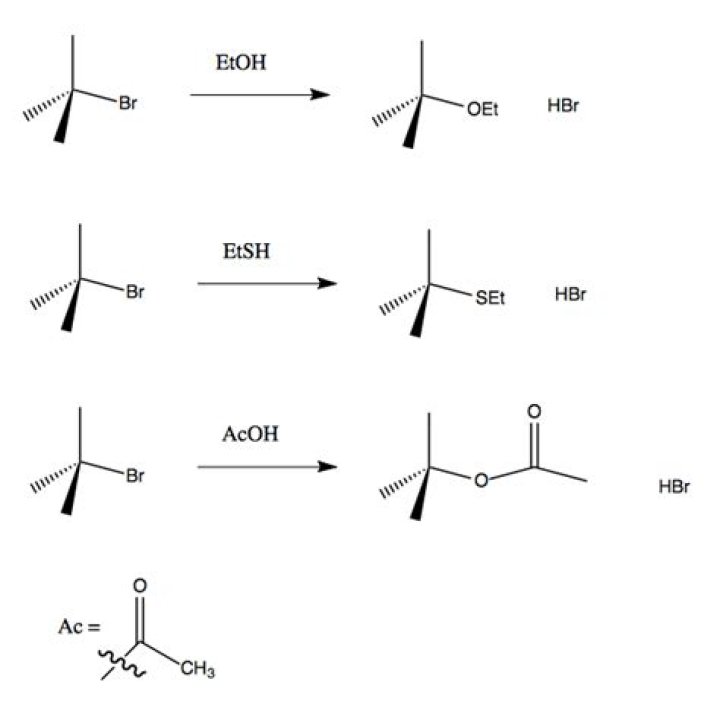
What is solvolysis ethanol?
An example of a solvolysis reaction is the reaction of a triglyceride with a simple alcohol such as methanol or ethanol to give the methyl or ethyl esters of the fatty acid, as well as glycerol. This reaction is more commonly known as a transesterification reaction due to the exchange of the alcohol fragments.
Which will undergo solvolysis in aqueous ethanol fastest?
Tertiary alkyl undergoes solvolysis in either acetic acid or in ethanol. In which solvent reaction is more rapid and why? Tertiary alkyl halide undergoes solvolysis in either acetic acid or in ethanol .
What is solvolysis explain with example?
solvolysis, a chemical reaction in which the solvent, such as water or alcohol, is one of the reagents and is present in great excess of that required for the reaction. The solvents act as or produce electron-rich atoms or groups of atoms (nucleophiles) that displace an atom or group in the substrate molecule.
What is solvolysis used for?
Solvolysis Process refers to a thermochemical process leading to depolymerization of UP composites, using hot water as a reactant.
What is solvolysis and hydrolysis?
is that hydrolysis is (chemistry) a chemical process of decomposition involving the splitting of a bond and the addition of the hydrogen cation and the hydroxide anion of water while solvolysis is (chemistry) any reaction, between a solute and its solvent, in which one or more bonds of the solute are broken; …
Which one of the following alkyl bromide undergoes most rapid solvolysis in methanol?
Option 1 (1-bromo-3-methylbut-2-ene) will go most rapid solvolysis in methanol solution to give corresponding methyl ether.
What are tertiary alkyl halides?
Tertiary alkyl halide (3o alkyl halide; tertiary haloalkane; 3o haloalkane): An alkyl halide (haloalkane) in which the halogen atom (F, Cl, Br, or I) is bonded to a tertiary carbon. Tert-butyl chloride (2-chloro-2-methylpropane), a typical tertiary alkyl halide.
What is solvolysis in organic chemistry?
Solvolysis: A reaction in which the solvent is a reactant, and becomes part of the reaction product. Hydrolysis of tert-butyl chloride; solvent = water.
What type of reaction is solvolysis?
In general, solvolysis is a type of substitution or elimination reaction in which the solvent acts as a nucleophile. In a solvolysis reaction there is usually an alkyl halide (an organic substrate containing a halogen) that acts as the electrophile and a solvent molecule that acts as the nucleophile.
What undergoes solvolysis in methanol most rapidly?
Which one of the following alkylbromides undergoes most rapid solvolysis in methanol solution to give corresponding methyl ether? Tertiary alkyl undergoes solvolysis in either acetic acid or in ethanol.