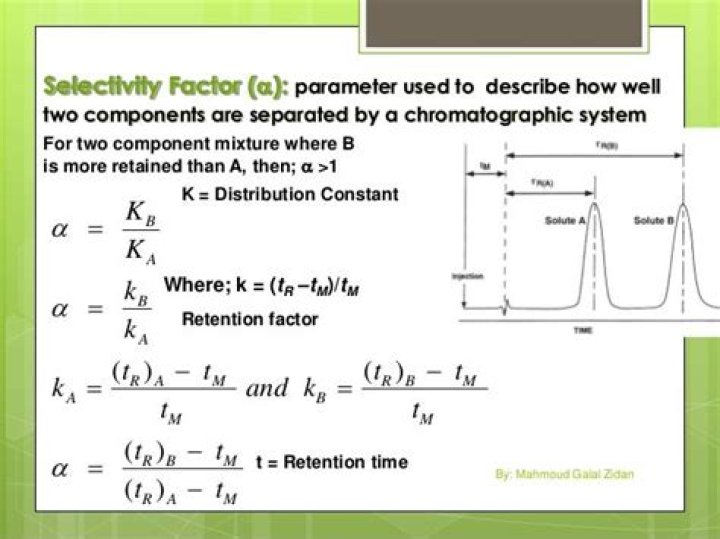
What is selectivity factor in chromatography?
The selectivity (or separation) factor (α) is the ability of the chromatographic system to ‘chemically’ distinguish between sample components. It is usually measured as a ratio of the retention (capacity) factors (k) of the two peaks in question and can be visualized as the distance between the apices of the two peaks.
What is selectivity factor in analytical chemistry?
Since all analytes spend the same amount of time in the mobile phase (equal to the dead time t0), selectivity is the ratio of the amount of time the later eluting analyte spends in the stationary phase relative to that of the earlier eluting analyte.
What must be the value of the selectivity factor?
Explanation: Selectivity factor/ Relative retention must always be greater than 1. It is also known as Relative migration rate.
What factors affect selectivity?
The selectivity series observed in ion chromatography seems to be best explained by the interplay of two components: electrostatic attraction (ES) and the enforced-pairing (EP) that is brought about by hydrophobic attraction and by water-enforced ion pairing.
What does a low selectivity factor mean?
Selectivity factor is a quantifiable measure of how efficient an antibiotic is during the process of gene selection. A selectivity factor lower than 10 means the concentration of antibiotic needed for selection is too close to the toxic concentration for the transfected cells.
How is selectivity calculated in HPLC?
Selectivity is the ability of an HPLC method to separate two analytes from each other. Selectivity usually is abbreviated with the Greek letter α, and is calculated as: α = k2 / k1 where k1 and k2 are the retention factors, k, of the first and second peaks of a peak pair.
What is selectivity in analytical method?
The recommended definition of Selectivity is: Selectivity refers to the extent to which the method can be used to determine particular analytes in mixtures or matrices without interferences from other components of similar behavior.
What is a good selectivity factor HPLC?
A good selectivity for HPLC is 1.1, which allows a resolution of 1.5 to be achieved with about 10,000 theoretical places. The critical pair in a separation is defined as adjacent solutes that have the smallest α value.
Is selectivity factor a good indicator of resolution?
The most important factor that influences resolution during a chromatographic separation is selectivity.
What selectivity means?
selectivity. / (sɪˌlɛkˈtɪvɪtɪ) / noun. the state or quality of being selective. the degree to which a radio receiver or other circuit can respond to and separate the frequency of a desired signal from other frequencies by tuning.
What 3 factors affect the selectivity of ion exchangers?
The factors that affect separation during ion exchange chromatography include the surface area of the stationary phase (resin bead size); the density of exchange sites on the stationary phase surface (cross-linkage); the flow rate of the mobile phase (resin bead size and column geometry; system pressure in high- …
What makes a good stationary phase?
Typically, the stationary phase is a porous solid (e.g., glass, silica, or alumina) that is packed into a glass or metal tube or that constitutes the walls of an open-tube capillary. The mobile phase flows through the packed bed or column.