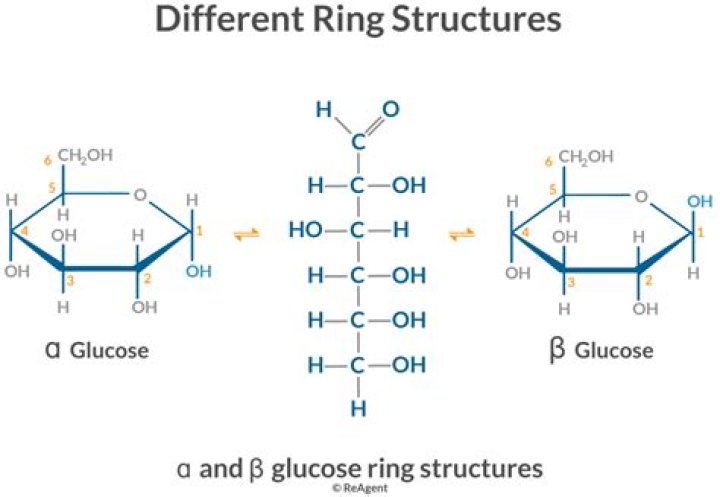
What is ring structure of glucose?
Glucose is a group of carbohydrates which is a simple sugar with a chemical formula C6H12O6. It is made of six carbon atoms and an aldehyde group. Therefore, it is referred to as an aldohexose. It exists in two forms viz open-chain (acyclic) form or ring (cyclic) form.
What is the structure of a glucose?
Glucose is defined as a group of carbohydrates, a simple sugar having a chemical formula C6H12O6. It is composed of six carbon atoms, including an aldehyde group. Thus, we can refer to this as an aldohexose. It exists in two forms, which are either in the open-chain (acyclic) form or ring (cyclic) form.
What is a glucose model?
Glucose Model. Glucose has a chemical formula of: C6H12O6 That means glucose is made of 6 carbon atoms, 12 hydrogen atoms and 6 oxygen atoms. You will be building one type of sugar called glucose. Glucose is produced during photosynthesis and acts as the fuel for many organisms.
Why is glucose shaped like a hexagon?
Glucose is a little hexagon made up of six carbon atoms and fructose is a pentagon of carbon atoms, and both have oxygen and hydrogens hanging off the hexagon and pentagon. They link with each other via an oxygen and make the sucrose molecule.
What are ring structures?
A Ring Structure is a cyclic compound that is a hydrocarbon in which the carbon chain joins to itself in a ring, and has atoms of at least two different elements as members of its ring(s).
Why is glucose a ring structure?
Ring Structure for Glucose: Due to the tetrahedral geometry of carbons that ultimately make a 6 membered stable ring , the -OH on carbon #5 is converted into the ether linkage to close the ring with carbon #1. This makes a 6 member ring – five carbons and one oxygen.
What is the structure and function of glucose?
Glucose (from Greek glykys; “sweet”) has the molecular formula C6H12O6. It is found in fruits and honey and is the major free sugar circulating in the blood of higher animals. It is the source of energy in cell function, and the regulation of its metabolism is of great importance (see fermentation; gluconeogenesis).
Why is glucose Dextrorotatory?
Glucose is by far the most common carbohydrate and classified as a monosaccharide, an aldose, a hexose, and is a reducing sugar. It is also known as dextrose, because it is dextrorotatory (meaning that as an optical isomer is rotates plane polarized light to the right and also an origin for the D designation.
What molecule has a ring structure?
Aromatic rings (also known as aromatic compounds or arenes) are hydrocarbons which contain benzene, or some other related ring structure. Benzene, C6H6, is often drawn as a ring of six carbon atoms, with alternating double bonds and single bonds: This simple picture has some complications, however.
How do you structure glucose?
- 4 Simple Steps – Chain Glucose Molecule.
- Draw 6 carbon molecules and draw arms except for the first one.
- Draw 5 hydrogen to carbon bonds.
- Fill remaining spaces with ( OH ) group.
- Complete the top end with hydrogen bonds and 1 oxygen double bond.
- 9 thoughts on “4 Simple Steps – Chain Glucose Molecule”