What is reagent acetic acid?
Acetic acid is a chemical reagent for the production of chemical compounds. The largest single use of acetic acid is in the production of vinyl acetate monomer, closely followed by acetic anhydride and ester production. The volume of acetic acid used in vinegar is comparatively small.
What is use of acetic acid?
Industrially, acetic acid is used in the preparation of metal acetates, used in some printing processes; vinyl acetate, employed in the production of plastics; cellulose acetate, used in making photographic films and textiles; and volatile organic esters (such as ethyl and butyl acetates), widely used as solvents for …
What is glacial acetic acid used for?
Diluted, glacial acetic acid is used in preservation and canning of pickles and other vegetables. It is also used as a descaling agent for cleaning. Along with cleaning and canning, diluted glacial acetic acid can treat or prevent bacterial or fungal outer ear infections.
Why is it called glacial acetic acid?
Acetic acid that contains a very low amount of water (less than 1%) is called anhydrous (water-free) acetic acid or glacial acetic acid. The reason it’s called glacial is that it solidifies into solid acetic acid crystals just cooler than room temperature at 16.
What is acetic acid made from?
Most acetic acid is made by methanol carbonylation, where methanol and carbon monoxide react to produce acetic acid. The compound is miscible with ethanol, ethyl ether, acetone, and benzene, and is soluble in carbon tetrachloride and carbon disulfide.
Why is acetic acid in vinegar?
Vinegar is essentially a dilute solution of acetic (ethanoic) acid in water. Acetic acid is produced by the oxidation of ethanol by acetic acid bacteria, and, in most countries, commercial production involves a double fermentation where the ethanol is produced by the fermentation of sugars by yeast.
What is the pH of acetic acid?
So, now we know that a 1 M acetic acid solution has a pH of 2.38.
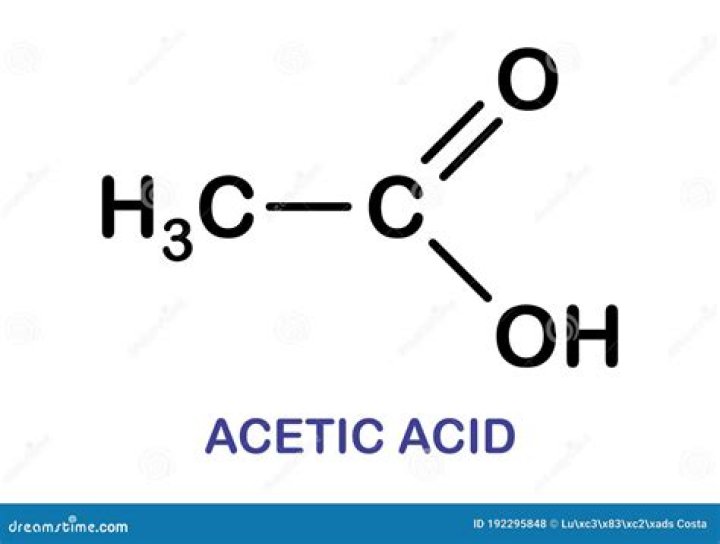
What is acetic acid formula?
CH₃COOH
Acetic acid/Formula
Is it safe to drink glacial acid?
Glacial acetic acid is a dangerous chemical that has been associated with several adverse drug events involving patients over recent years. When diluted to the proper concentration, acetic acid solutions have a variety of medicinal uses.
How do you dispose of glacial acetic acid?
11). Burn in a chemical incinerator equipped with an afterburner and scrubber but exert extra care in igniting as this material is highly flammable. Offer surplus and non- recyclable solutions to a licensed disposal company. Contact a licensed professional waste disposal service to dispose of this material.
What is the pH of glacial acetic acid?
Glacial acetic acid (a saturated solution at around 17 M) has a pH of 2.4.
What is difference between acetic acid and glacial acetic acid?
Acetic acid is a solution of acetic acid and water. Glacial acetic acid is nothing but the concentrated form of acetic acid solution. Acetic acid contains more water and is in its diluted form. Glacial acetic acid has less than 1% of water content and is concentrated.
How do you convert acetic acid to ethanol in organic chemistry?
Organic chemistry. Reduction of acetic acid gives ethanol. The OH group is the main site of reaction, as illustrated by the conversion of acetic acid to acetyl chloride. Other substitution derivatives include acetic anhydride; this anhydride is produced by loss of water from two molecules of acetic acid.
What reagent can be used to form a carboxylic acid?
Carboxylic acid (COOH, CO 2 H) can also be formed as well except it is through an oxidative workup step instead. H 2 O 2 (hydrogen peroxide) is the reagent to look out for because as seen below it turns: – Ends of alkenes with 1 –H ≠ Aldehydes but Carboxylic Acids instead
What is the reaction between ethanol and ethanoic acid?
Or, ethanol and ethanoic acid gives ethyl ethanoate + water. Most acetate esters, however, are produced from acetaldehyde using the Tishchenko reaction. In addition, ether acetates are used as solvents for nitrocellulose, acrylic lacquers, varnish removers, and wood stains.