What is meant by supercritical fluid chromatography?
What is meant by supercritical fluid chromatography?
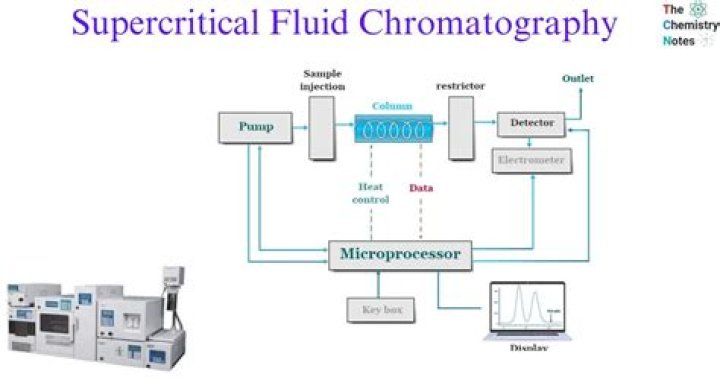
Supercritical fluid chromatography (SFC) is a separation technique similar to gas and liquid chromatography, but using a supercritical fluid as mobile phase. If a liquid or a gas is used above its critical temperature and pressure, it changes to a supercritical fluid.
What is supercritical fluid method?
Supercritical fluid extraction (SFE) is the process of separating one component (the extractant) from another (the matrix) using supercritical fluids as the extracting solvent. Extraction is usually from a solid matrix, but can also be from liquids. These essential oils can include limonene and other straight solvents.
What is the difference between HPLC and SFC?
SFC offers a higher performance than HPLC and often shorter equilibration times ‘*16, because following changes of the super- critical fluid density a stable system is rapidly obtained. From the above it can be concluded that HPLC requires the longest method development time.
Why is SFC better than HPLC?
Various types of packed columns can be used, including silica or bonded silica; alternatively, fused silica capillary columns can be used (Raynor et al., 1988). A major advantage of SFC over HPLC is the shorter retention times; compounds elute as sharp peaks and the sensitivity of detection is accordingly increased.
Why is SFC better than GC and LC?
The physical properties of supercritical fluids between liquids and gases enables the SFC technique to combine with the best aspects of HPLC and GC, as lower viscosity of supercritical fluids makes SFC a faster method than HPLC. Lower viscosity leads to high flow speed for the mobile phase.
What is subcritical CO2 extraction?
Subcritical (low temperature, low pressure) CO2 extractions take more time and produce much smaller yields than supercritical, but they keep essential oils, terpenes and other sensitive chemicals in the plant.
What is the importance of supercritical fluid extraction?
Major benefits of supercritical fluid extraction is the ability to control the extraction parameters like, temperature and pressure which has a direct impact on fluid density, improved transport properties, higher extraction yield and shorter extraction time.
What is subcritical fluid?
Subcritical water is liquid water under pressure at temperatures above usual boiling point, 100 °C (212 °F). It is also known as “subcritical water” or “pressurized hot water.” At subcritical state, water is maintained in liquid form by apply pressure.
What is supercritical condition?
The supercritical condition of a steam-water cycle is a state at which its temperature and pressure are above its thermodynamic critical point, where the pressure of the steam water is 22.12 MPa, the temperature is 647.14 K, and the density is 324 kg/m3.
Why co2 is used in SFC?
Mobile Phases There is a wide variety of materials used as mobile phase in SFC. The most common supercritical fluid which is used in SFC is carbon dioxide because its critical temperature and pressure are easy to reach.
Why is SFC faster than HPLC?
SFC is theoretically up to ten times faster than HPLC,7 because of the lower viscosity and higher diffusivity in the mobile phase, SFC columns typically provide a three- to five-fold reduction in analysis time over HPLC.