What is meant by alicyclic compounds?
alicyclic compound, in chemistry, any of a large class of organic compounds in which three or more atoms of the element carbon are linked together in a ring. In the larger rings all the bond angles have the preferred value (about 109.5°); consequently, the atoms in the ring do not lie in one plane.
Are cyclic compounds more stable?
According to the Bayer Theory, cyclopentane would be the most stable because its bond angles, 108°, are closest to the ideal angle of 109.5°. Furthermore, cyclic compounds do not become less and less stable as the number of rings increases.
What are the difference between alicyclic and aliphatic?
Aliphatic compounds have single or multiple carbon bonds but do not have cyclic structures. Alicyclic compounds are formed by joining two carbon atoms of an aliphatic chain through a covalent bond resulting in a cyclic structure.
What is alicyclic compound with example?
Generally, alicyclic compounds resemble analogous aliphatic compounds. An example of an alicyclic compound is: Cyclohexane (C6H12) and so are many terpenes, like menthol. A compound that contains at least one aromatic ring is defined as Aromatic compounds.
What are some examples of alicyclic compound?
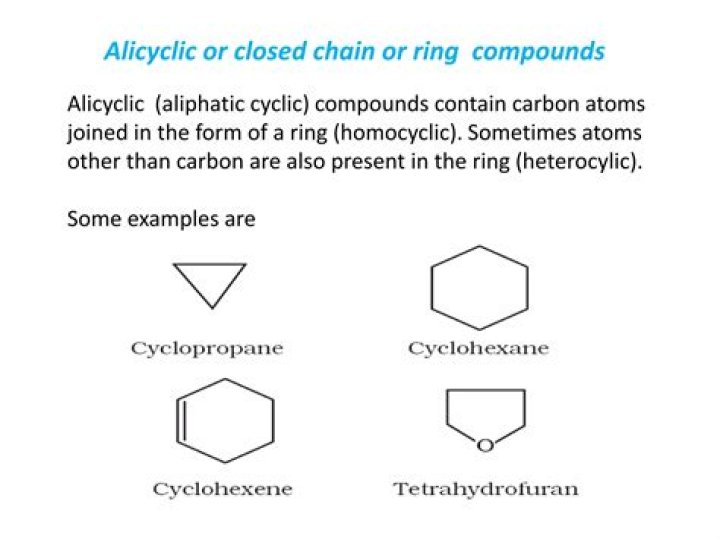
The simplest alicyclic compounds are the monocyclic cycloalkanes: cyclopropane, cyclobutane, cyclopentane, cyclohexane, cycloheptane, cyclooctane, and so on. Bicyclic alkanes include bicycloundecane, decalin, and housane. Polycyclic alkanes include cubane, basketane, and tetrahedrane.
Are cycloalkenes saturated?
Cycloalkanes are saturated hydrocarbons that contain a ring in their carbon backbones. Analogous ring structures containing double and triple bonds are known as cycloalkenes and cycloalkynes.
What are the limitations of Baeyer strain theory?
Limitations of Baeyer Strain Theory in Cycloalkanes According to Baeyer, larger ring structures are not feasible due to negative pressure, but they do exist and are very stable. To remove angle pressure, larger ring structures are wrinkled (puckered) rather than planar.
Are alicyclic compounds heterocyclic?
When the ends of the chains are joined together into a ring, cyclic compounds result; such substances often are referred to as carbocyclic or alicyclic compounds. Substitution of one or more of the ring carbon atoms in the molecules of a carbocyclic compound with a heteroatom gives a heterocyclic compound.
What is alicyclic and aliphatic compounds?
Answer: An alicyclic compound is defined as an organic compound, which is both cyclic and aliphatic. They either have one or more all-carbon rings that may be either saturated or unsaturated, but they do not hold aromatic character. Alicyclic compounds can have either one or more aliphatic side chains attached.
Are all aromatic compounds alicyclic?
Aromatic compounds are also cyclic compounds having an enclosed ring structure. However, the difference between alicyclic and aromatic compounds is that alicyclic compounds do not have a delocalized pi electron cloud whereas aromatic compounds are essentially composed of a delocalized pi electron cloud.
What are alicyclic and aromatic compounds define with examples?
An organic compound in which carbon atoms are linked to form one or more rings. Aromatic compounds are excluded because of their special properties. In general, alicyclic compounds resemble analogous aliphatic compounds. Example——-Cyclohexan (C6H12) is alicyclic, as are manyterpenes, such as menthol.