What is difference between alkanes alkenes and alkynes?
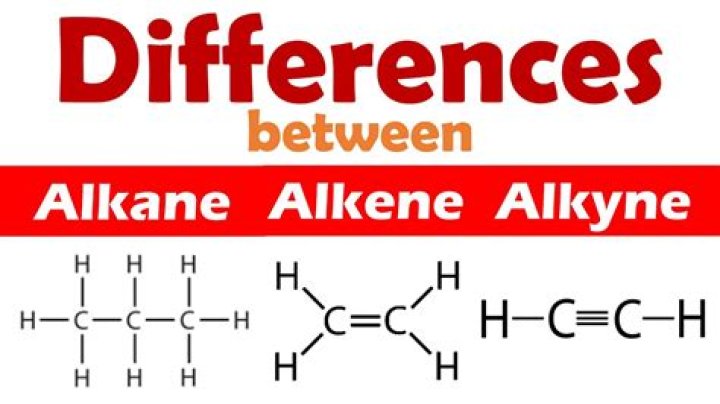
What is difference between alkanes alkenes and alkynes?
The main difference between alkanes, alkenes, and alkynes is that alkanes form a single bond between carbon atoms while alkenes form a double bond while alkynes form a triple bond between the carbon atoms. Alkanes are also known as paraffin. They are unsaturated hydrocarbons because of the presence of a double bond.
What are alkanes alkenes alkynes Class 10?
Alkanes are the simplest hydrocarbons known to us. They have a general formula of CnH2n+2. Alkanes belong to the family of saturated hydrocarbons that is; they contain only sigma bond linkages between carbon and hydrogen. Alkenes and alkynes, on the other hand, are unsaturated hydrocarbons.
What are the 4 alkenes?
13.1: Alkenes- Structures and Names
| IUPAC Name | Molecular Formula | Melting Point (°C) |
|---|---|---|
| ethene | C2H4 | –169 |
| propene | C3H6 | –185 |
| 1-butene | C4H8 | –185 |
| 1-pentene | C5H10 | –138 |
How are alkynes made?
The loss of a hydrogen atom and a halogen atom from adjacent alkane carbon atoms leads to the formation of an alkene. The loss of additional hydrogen and halogen atoms from the double‐bonded carbon atoms leads to alkyne formation.
How do you know if alkane is alkene or alkyne?
Explanation:
- Alkanes have single bonds between carbons in a hydrocarbon. A quick way to recognize an alkane is the general formula: CnH2n+2 .
- Alkenes have double bonds between carbons. A quick way to recognize an alkene is its general formula: CnH2n .
- And alkynes have triple bonds between carbons.
Why are alkynes more reactive than alkenes?
Alkynes > Alkenes > Alkanes. Alkynes have two pi-bonds between two (or more) carbon atoms, along with a sp-sp hybridised orbital bonding (sigma bond). The pi-bonds can be easily broken to free the valence shell electrons for combining with other atoms. Hence they are the most reactive.
Which is named first alkene or alkyne?
Higher alkenes and alkynes are named by counting the number of carbons in the longest continuous chain that includes the double or triple bond and appending an -ene (alkene) or -yne (alkyne) suffix to the stem name of the unbranched alkane having that number of carbons.
What are alkanes alkenes and alkynes give one example of each?
→ Examples : Ethylene ( C2H2 ) or ethene; Pentene ( C5H10 ). Alkynes are unsaturated hydrocarbons which have atleast one carbon- carbon triple bond. Their general formula is CnH2n−2 . You can see that these have 2 hydrogen atoms less than their corresponding alkane.