What is conformation with example?
Conformation can be defined as the shape adopted by a molecule caused by the rotation around one or more single bonds. For instance, in the case of alkanes, there is a distribution of electrons around the internuclear axis of the C-C bond.
How many conformational isomers are there?
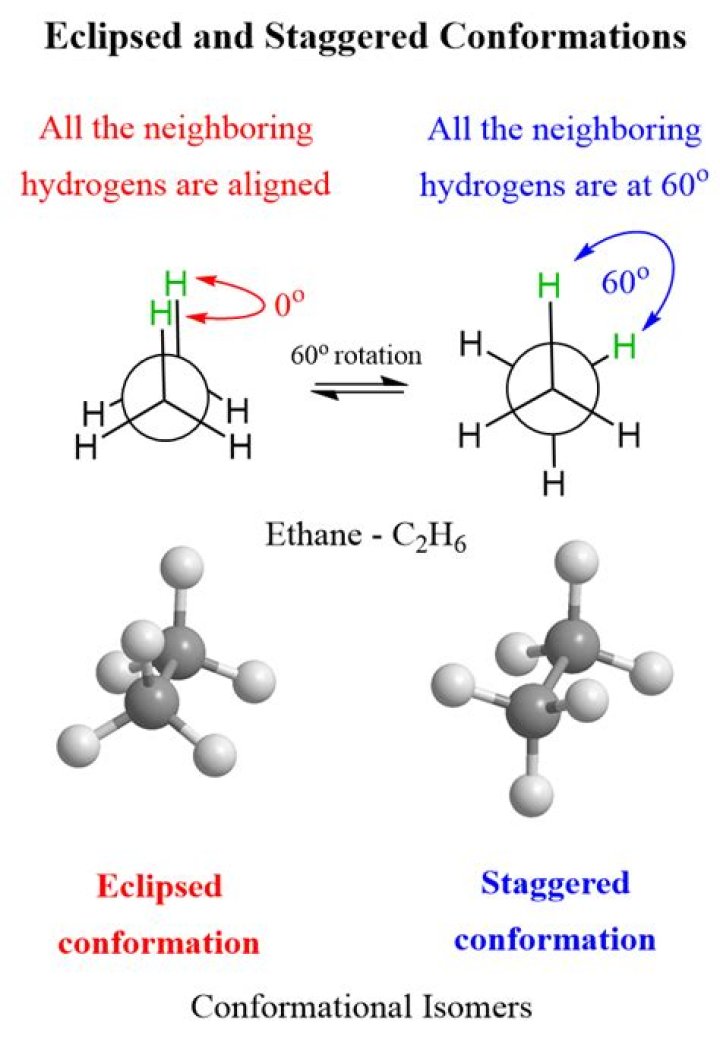
There are two types of conformational isomers: -Eclipsed conformational isomers: In these isomers, the carbons are aligned so that the hydrogens are lined up with each other.
Are conformational isomers isomers?
Conformational isomers (or conformers or rotational isomers or rotamers) are stereoisomers produced by rotation (twisting) about σ bonds, and are often rapidly interconverting at room temperature.
What are the types of conformations?
Conformational Isomers
- Eclipse conformation. Conformation in which hydrogen atoms attached to two carbons areas nearest to each other as possible is known as eclipsed.
- Staggered conformation.
What are three types of conformational isomers?
A: antiperiplanar, anti or trans. B: synclinal or gauche. C: anticlinal or eclipsed. D: synperiplanar or cis.
Does ethane have an isomer?
Therefore, ethane, like methane, really has only one form. If substitutions are made in the ethane molecule—for instance, exchanging some of the hydrogen atoms for deuterium atoms to make 1,2-dideuterioethane—isomeric staggered forms become possible.
Are conformations isomers?
While any two arrangements of atoms in a molecule that differ by rotation about single bonds can be referred to as different conformations, conformations that correspond to local minima on the potential energy surface are specifically called conformational isomers or conformers.
Is a ring flip a conformational isomer?
In methylcyclohexane, for example, there are two conformational isomers, one with the methyl group axial and one with the methyl group equatorial. The two interconvert through ring flipping.
What do you mean by conformations?
conformation, any one of the infinite number of possible spatial arrangements of atoms in a molecule that result from rotation of its constituent groups of atoms about single bonds.
Is cyclopentane an isomer of pentane?
Cyclopentane is not an isomer of pentane because it has only 10 hydrogen atoms where pentane has 12.
Is ch4 a isomer?
Chemical compounds that have the same molecular formula, but different molecular structures are said to be isomers….3.3. 2 Alkanes.
| Compound name | Molecular formula | Number of isomers |
|---|---|---|
| Methane | CH4 | 1 |
| Ethane | C2H6 | 1 |
| Propane | C3H8 | 1 |
| Butane | C4H10 | 2 |