What is Chloromethylation process?
The Blanc chloromethylation (also called the Blanc reaction) is the chemical reaction of aromatic rings with formaldehyde and hydrogen chloride catalyzed by zinc chloride or other Lewis acid to form chloromethyl arenes. The reaction was discovered by Gustave Louis Blanc (1872-1927) in 1923.
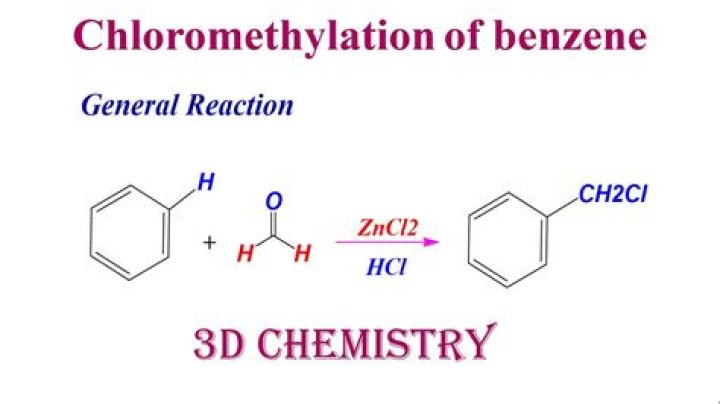
Which reagent is used for Chloromethylation of benzene?
In the reaction, we have been given benzene as the substrate and the reagents are formaldehyde and hydrochloric acid. This reaction is chloromethylation of benzene. The benzene reacts with formaldehyde in the presence of HCl to give benzene methyl chloride. It is an electrophilic substitution reaction.
Which of the following reagents use chloro methylation?
1. Zinc chloride was found to catalyze chloromethylation of resins for solid-phase peptide synthesis, producing low levels (0.07–1.27 mmol/g) of chloromethylation more accurately and under more convenient conditions than SnCl4. 2.
How is bis chloromethyl ether formed?
It is also produced as a byproduct in the Blanc chloromethylation reaction, formed when formaldehyde (the monomer, paraformaldehyde or formalin) and concentrated hydrochloric acid are mixed, and is a known impurity in technical grade chloromethyl methyl ether.
How is thiophene made?
i) Thiophene can be synthesized on industrial scale by the high temperature reaction between n- butane and Sulfur. ii) Thiophene can be synthesized by passing a mixture of acetylene and hydrogen sulfide through a tube containing alumina at 400°C. This method is commercially used.
What is paraformaldehyde used for?
Paraformaldehyde can be used as a substitute of aqueous formaldehyde to produce the resinous binding material, which is commonly used together with melamine, phenol or other reactive agents in the manufacturing of particle board, medium density fiberboard and plywood.
When thiophene react with Hcho HCL what product is formed?
Chloromethylation and formylation : Thiophene reacts with formaldehyde and hydrochloric acid to give 2 – chloro methyl thiophene.
Which of the following reagents are used in Balz Schiemann reaction?
The reactants that take part in the Balz Schiemann reaction are aromatic amines, nitrous acid, and fluoroboric acid. The Schiemann reaction is a chemical reaction in which the main aromatic amine is converted into a chemical compound famously known as aryl fluoride.
Which of the following reagents can be used to prepare salicylaldehyde from phenol?
Salicylaldehyde is prepared by heating phenol and chloroform together in the presence of potassium hydroxide and sodium hydroxide. This reaction is called the Relmer-Tiemann reaction. The correct option is Option C, Reimer-Tiemann reaction.
What is BIS methyl?
Bis(chloromethyl) ether is a clear liquid with a strong unpleasant odor. Bis(chloromethyl) ether is an ether that is dimethyl ether in which one of the hydrogens attached to each of the methyl group has replaced by a chlorine. It has a role as a carcinogenic agent and an alkylating agent.
What functional group is thiophene?
Thiophene is a heterocyclic compound with the formula C4H4S. Consisting of a planar five-membered ring, it is aromatic as indicated by its extensive substitution reactions. It is a colorless liquid with a benzene-like odor.