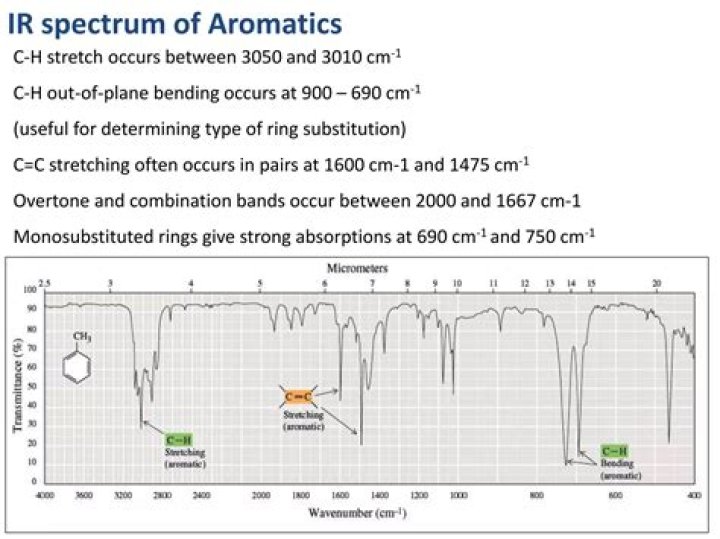
What is aromatic CH stretch?
The =C–H stretch in aromatics is observed at 3100-3000 cm-1. This is a very useful tool for interpreting IR spectra: Only alkenes and aromatics show a C–H stretch slightly higher than 3000 cm-1. Compounds that do not have a C=C bond show C–H stretches only below 3000 cm-1.
What is sp3 CH stretch?
The sp3 C-H stretching vibrations are just to the right of 3000 cm-1 and the sp2 C-H just to the left of 3000 cm-1. b. A cis-alkene, cis-pent-2-ene. The sp3 C-H stretching vibrations are just to the right of 3000cm-1 and since it is an internal alkyne, there is no sp C-H near 3300cm-1. 3b.
What effect does conjugation have on the location of C O peaks?
Conjugation weakens the C=O. bond, lowers its force constant, and hence its peak position is lowered, on average about 30 cm-1 (1).
Where does C C show up on IR?
| Functional Group | Frequency (cm-1) | intensity |
|---|---|---|
| C-OH stretch | 1200-1020 | strong |
| NO2 stretch | 1600-1500 and 1400-1300 | strong |
| C-F | 1400-1000 | strong |
| C-Cl | 800-600 | strong |
What are stretching frequencies?
Stretching frequencies are higher than corresponding bending frequencies. (It is easier to bend a bond than to stretch or compress it.) Bonds to hydrogen have higher stretching frequencies than those to heavier atoms.
What is an sp2 C-H?
Ch 2: sp2 hybridisation. Chapter 2 : Alkanes. sp2 hybridisation. When a C atom is attached to 3 groups and so is involved in 3 σ bonds, it requires 3 orbitals in the hybrid set. This requires that it is sp2 hybridised.
What is the IR frequency for alkane C-H stretching?
The C-H-stretching modes can be found between 2850 and 3300 cm-1, depending on the hydrization. The range from 2850-3000 cm-1 belongs to saturated systems (alkanes, sp3, example 1), while the peaks from 3000-3100 cm-1 indicate an unsaturated system (alkenes, sp2, example 2; aromatic ring, example 3,4).
How does conjugation influence the stretching frequency of double bond?
Conjugation with a double bond or benzene ring lowers the stretching frequency. The 30 to 40 cm-1 decrease in frequency is illustrated by the following examples. The stretching frequency of the conjugated double bond is also lowered (blue notation) and may be enhanced in intensity.
How does conjugation affect vibrational frequency?
Conjugation increases the single bond character of C=C and C=O Bonds in the resonance structure. Two bands are usually seen for N=O stretching frequency due to symmetric and Asymmetric stretching.
What is a stretch in IR spectroscopy?
When there is a continuous change in the interatomic distance along the axis of the bond between two atoms, this process is known as a stretching vibration. A change in the angle occurring between two bonds is known as a bending vibration. Four bending vibrations exist namely, wagging, twisting, rocking and scissoring.