What is anti capillary action?
Capillary action is the phenomenon of a liquid flowing in narrow spaces without the assistance of, and in opposition to external forces like gravity. Anti-capillary (water blocked) wire and cable. Our water blocked product consists of bare or tinned copper conductors sealed with a silica blocking agent.
What is capillary action of water?
Capillary action is important for moving water (and all of the things that are dissolved in it) around. It is defined as the movement of water within the spaces of a porous material due to the forces of adhesion, cohesion, and surface tension.
How can capillary action stop water?
Wind pressure can also act on the water and drive it upwards even further. Incorporating a capillary break. by detailing a gap of 6 mm between surfaces will stop capillary action occurring, as the surfaces will be too far apart for water to bond between them.
What is capillary action example?
Water moving up in straw or glass tube against gravity, tears moving through tear ducts, water moving through a cloth towel against gravity. These are examples of capillary action.
Why does water go up a capillary tube?
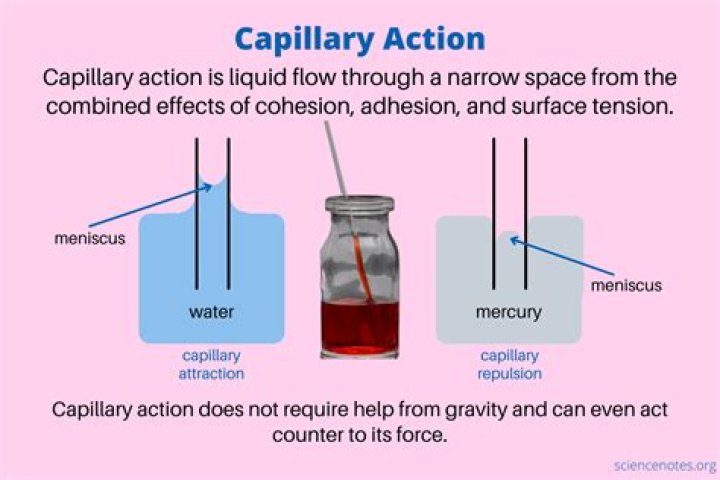
Water rises inside the capillary tube due to adhesion between water molecules and the glass walls of the capillary tube. This adhesion, together with surface tension in the water, produces an effect called capillarity , with a characteristic concave surface. The narrower the tube, the higher the water will rise.
How high will capillary action lift water?
Capillary action and root pressure can support a column of water some two to three meters high, but taller trees–all trees, in fact, at maturity–obviously require more force.
What is capillary rise of water?
DEFINITION: A rise in a liquid above the level of zero pressure due to a net upward force produced by the attraction of the water molecules to a solid surface, e.g. glass, soil (for those cases where the adhesion of the liquid to the solid is greater than the cohesion of the liquid to itself)
What is responsible for the capillary action of water quizlet?
Capillary action occurs because water is sticky, and, thanks to forces of cohesion and adhesion, are attracted. A process powered by adhesion that causes water molecules to move upward through a narrow glass tube.
How high can capillary action take water?
Capillary rise of liquid in a capillary Likewise, lighter liquid and lower gravity increase the height of the column. Thus for a 2 m (6.6 ft) radius glass tube in lab conditions given above, the water would rise an unnoticeable 0.007 mm (0.00028 in).
Does capillary action prevent water from moving through materials with pores?
Capillary action is due to the attraction among molecules of water and surrounding materials. Capillary action prevents water from moving through materials with pores. Capillary action causes clothing to stay dry. Since water is polar molecule they climb over and on each other and the pores gradually go upward.
What are the factors affecting the capillary rise of water?
For this study, the factors used to determine capillary rise are the diameter of the capillary tube (representing the diameter of the pores in a soil), the contact angle between the liquid and the surface to which it adheres, the density of the liquid, the viscosity of the liquid, surface tension, and whether or not …
Is capillary water available to plants?
It is not plant available. Capillary water (also called soil solution) is water in the micropores of the soil and is held against the force of gravity. Capillary water is held by cohesion and adhesion and most of it is plant available. Hygroscopic water is held very tightly on the surfaces of soil particles.