What is an iodate ion?
Iodate is an iodine oxoanion and a monovalent inorganic anion. It is a conjugate base of an iodic acid.
What is iodide ion?
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate.
What is the meaning of iodate?
iodated; iodating. Medical Definition of iodate (Entry 2 of 2) : to impregnate or treat with iodine.
What is the name for IO3?
IOPHENOXIC ACID
IO3 : Summary
| Code | IO3 |
|---|---|
| Molecule name | IOPHENOXIC ACID |
| Systematic names | Program Version Name ACDLabs 12.01 (2S)-2-(3-hydroxy-2,4,6-triiodobenzyl)butanoic acid OpenEye OEToolkits 1.7.0 (2S)-2-[(3-hydroxy-2,4,6-triiodo-phenyl)methyl]butanoic acid |
| Formula | C11 H11 I3 O3 |
| Formal charge | 0 |
What is the oxidation number of I2O5?
+5
There are two iodine atoms. The overall charge on the whole molecule is zero because it is a neutral molecule. Therefore, the oxidation state of iodine is calculated as shown below. Hence, the oxidation state of iodine in I2O5 I 2 O 5 is +5.
Is the iodate ion io3 an oxidizing or reducing agent?
Potassium iodate is an oxidizing agent and as such it can cause fires if in contact with combustible materials or reducing agents.
What is iodide used for?
Potassium iodide (KI) is a chemical compound that can be used to protect the thyroid gland from possible radiation injury caused by radioactive iodine (radioiodine). Some radiological emergencies may release large amounts of radioiodine to the environment.
Is IO3 a polar molecule?
Interestingly, among these six stoichiometrically identical compounds, α-(H3O)2Pt(IO3)6 is polar, whereas other compounds are nonpolar and centrosymmetric. The material, α-(H3O)2Pt(IO3)6, exhibits very strong second harmonic generation (SHG) effects, approximately 1.2 × KTiOPO4 (KTP), and is phase-matchable.
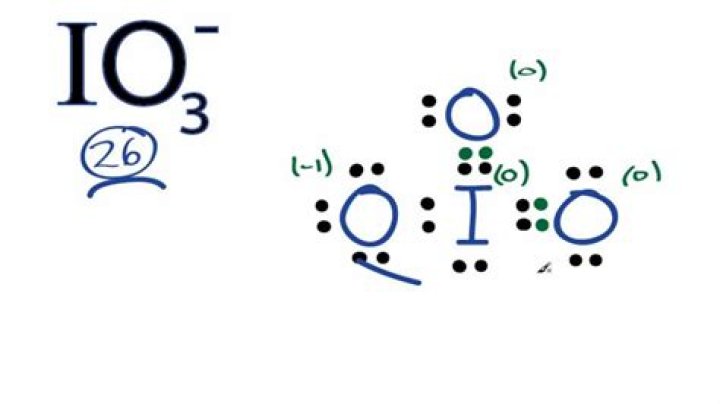
What is the shape of IO3?
Iodate is pyramidal in structure. The O–I–O angles range from 97° to 105°, somewhat smaller than the O–Cl–O angles in chlorate.