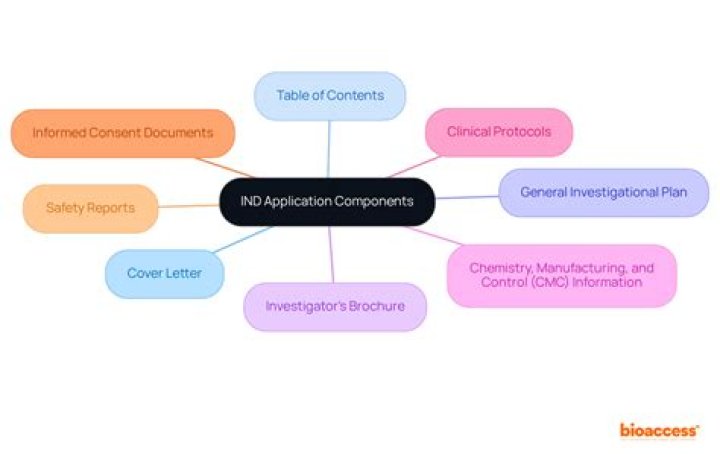
What is an IND study?
An Investigational New Drug Application (IND) is a request from a clinical study sponsor to obtain authorization from the Food and Drug Administration (FDA) to administer an investigational drug or biological product to humans.
What studies are needed for an IND?
What are IND-Enabling Studies?
- Pharmacodynamics and safety pharmacology.
- Pharmacokinetics, including absorption, distribution, metabolism, and excretion (ADME) and radiolabeled mass balance studies.
- Toxicology, including single-dose, repeated-dose, reproductive and developmental toxicity, and genotoxicity studies.
What does IND-enabling studies mean?
What is an Investigational New Drug or IND Program? The purpose of IND-enabling studies is to secure approval to conduct the first-in-human clinical trials with a new drug. To support IND-enabling studies, nonclinical (i.e., “non-human”) studies are conducted to evaluate the efficacy and safety of the drug.
What does IND mean in medical terms?
Investigational New Drug (IND) Application.
When can you file for IND?
The filing deadline is within 60 days of annual date of the IND. If there are multiple protocols under a single IND, each should be identified by title and have a summary report. The investigator should include the status of each study still in progress and each study completed during the previous year.
What is difference between IND and NDA?
The NDA is a formal request made by a Sponsor to market a new drug in the United States. Data collected during the IND phase of the development program will become part of the NDA. However, the NDA is much more comprehensive than the IND and is expected to provide very detailed information about the drug.
What are toxicology studies?
Toxicology Studies: Critical to Assessing Biologic Safety Toxicology studies are used to characterize the toxicity profile of a drug by identifying its impact on organ structure and / or functionality. This information is key in guiding the parameters for clinical trials to maximize safety and minimize risk.
How much do IND-enabling studies cost?
Yes, You Need Collaborators! Being Patience and Persistent! The only problem: it will cost $2-3 millions for IND-enabling studies and another $5-10M for a phase I trial.
How long does IND-enabling studies take?
Drug Safety and Metabolism (8-10 months) – Pilot pharmacokinetic & toxicology studies; – Drug safety IND-enabling toxicity studies(14-28day rodent and non-rodent); – Genotoxicity assays; – Safety pharmacology profile.
Are IND-enabling studies preclinical?
IND- and CTA-enabling studies comprise preclinical pharmacology, toxicology, metabolism and manufacturing studies that allow you to submit a formal application with either the FDA or EMA.
What is an IND nursing?
Abbreviation for investigational new drug.
How long do IND enabling studies take?