What is an example of reference electrode?
What is an example of reference electrode?
A reference electrode refers to an electrode that has an established electrode potential. In an electrochemical cell, the reference electrode can be used as a half cell. Some examples of reference cells include: Calomel electrode: This reference electrode consists of a mercury and mercury-chloride molecules.
Do you need a reference electrode?
The role of the reference electrode is to provide a stable potential for controlled regulation of the working electrode potential and in doing so allow the measurement of the potential at the working electrode without passing current through it. An ideal reference electrode should also have zero impedance.
What is the most common reference electrode?
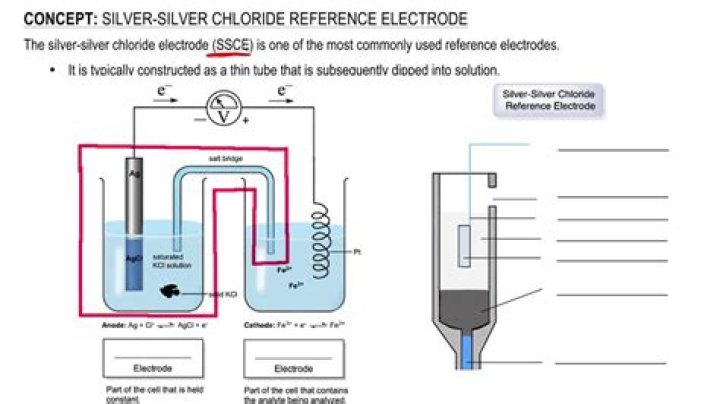
Silver/silver chloride reference electrodes
Silver/silver chloride reference electrodes are the most common type of reference electrode. They are very easy to manufacture and are able to function in a wide variety of ambient temperatures. The maximum temperature at which this type of electrode can function is about 130 degrees Celsius.
What are the different reference electrodes?
Aqueous reference electrodes Normal hydrogen electrode (NHE) (E ≈ 0.000 V) concentration H+ = 1 Molar. Reversible hydrogen electrode (RHE) (E = 0.000 V – 0.0591 × pH) Saturated calomel electrode (SCE) (E = +0.241 V saturated) Copper-copper(II) sulfate electrode (CSE) (E = +0.314 V)
What happens when there is no reference electrode?
Without reference electrode you can run a slow scan rate CV (typically 1 mV/s for a biofuel cell, depending on the size/surface area of your electrodes) from your OCV to 0 V (short circuit) with the configuration you proposed (with reference connection of your potentiostat connected to your counter electrode).
How do you use reference electrodes?
Testing Procedure
- Partially fill a beaker with electrolyte.
- Immerse the tip of your Reference Electrode into the solution.
- Add a high surface area platinum wire or graphite rod counter electrode to the solution.
- Connect the Reference Electrode to the Working (green) and Working Sense (blue) leads of your potentiostat.
How many types of reference electrodes are there?
What is the difference between reference electrode and indicator electrode?
The key difference between indicator electrode and reference electrode is that indicator electrode responds to changes in the activity of the analyte, whereas reference electrode does not respond to changes, and its response is stable.
Which of the following electrodes are used as a reference electrode?
The most commonly used reference electrode is calomel electrode.