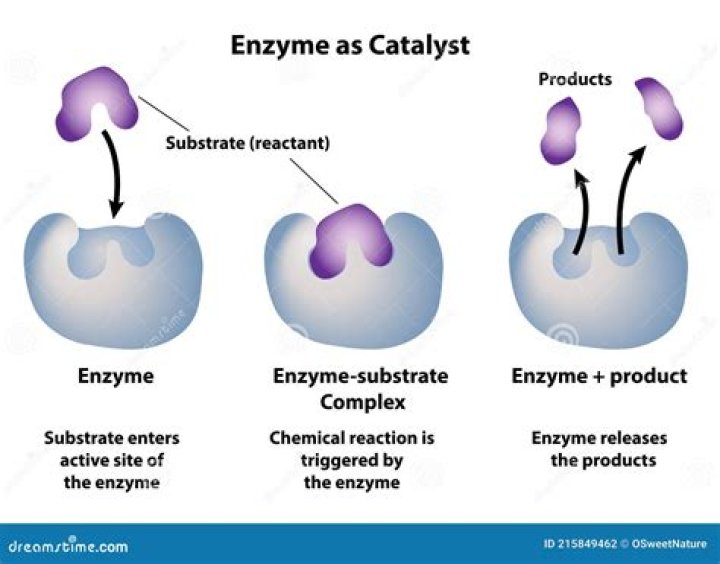
What is an example of a chemical catalyst?
These catalysts speed up chemical reactions but do not change their structure in the process. Examples of inorganic catalysts in chemical reactions include: potassium permanganate – Hydrogen peroxide will decompose into water and oxygen gas. A catalyst of potassium permanganate can be used to speed up this process.
What are common catalysts?
Here are five common chemical catalysts used within the manufacturing industry.
- Aluminosilicates. Aluminosilicates are a critical component of modern petrochemical manufacturing.
- Iron. Iron has long been the preferred catalyst for ammonia production.
- Vanadium.
- Platinum + Alumina.
- Nickel.
What are some examples of catalysts in everyday life?
Almost everything in your daily life depends on catalysts: cars, Post-It notes, laundry detergent, beer. All the parts of your sandwich—bread, cheddar cheese, roast turkey. Catalysts break down paper pulp to produce the smooth paper in your magazine. They clean your contact lenses every night.
What are catalysts in chemistry?
A catalyst is a substance that speeds up a chemical reaction, or lowers the temperature or pressure needed to start one, without itself being consumed during the reaction. Catalysis is the process of adding a catalyst to facilitate a reaction.
Is baking soda a catalyst?
Jekyll and Hyde Compound Chemical leavening requires an acidic catalyst in the batter, such as yogurt or buttermilk. On contact with the sodium bicarbonate, this causes the release of carbon dioxide in a simple acid-base reaction.
What are the best catalysts?
Metals like platinum and nickel make good catalysts because they adsorb strongly enough to hold and activate the reactants, but not so strongly that the products can’t break away. The simplest example of this is the reaction between ethene and hydrogen in the presence of a nickel catalyst.
Where are catalysts used?
Catalysts are used in industries to break down pulp to produce sanitary paper, to turn milk into yogurt, and to refine crude oil into a series of end products, among countless other uses. When a catalyst is used, a polluting chemical reaction can be reduced or replaced with an environment-friendly one.
Is Salt a catalyst?
Salt can be considered a catalyst in the reaction but has a different role than most catalysts. Copper II sulfate and aluminum react very slowly because aluminum is coated with a very thin layer of tarnish (aluminum oxide). This reaction can be sped up if the layer of aluminum oxide is removed or compromised.
Is vinegar a catalyst for baking soda?
If we use more vinegar, then there will be more for the baking soda to react with. However, using the best ratio of vinegar to baking soda will still give you a slower reaction than hydrogen peroxide with yeast, since the vinegar and baking soda reaction does not use a catalyst.
Is dish soap a catalyst?
In this experiment we add dish soap, to help create bubbles, and a catalyst — which is a special chemical that allows us to fast forward a reaction.